Chronic spontaneous urticaria is a prevalent and difficult-to-treat condition that has a very negative impact on patient quality of life.
ObjectiveTo describe the epidemiological and clinical characteristics of patients presenting with chronic spontaneous urticaria and the response to treatment administered according to the EAACI/GA(2)LEN/EDF/WAO consensus guideline.
Material and methodDescriptive cross-sectional study of all the patients with chronic spontaneous urticaria who consulted a skin allergy unit in the dermatology department of a tertiary hospital in Spain between July 2011 and July 2015.
ResultsThe study included 100 patients with chronic spontaneous urticaria; inducible urticaria was present in 45% of cases, and angioedema in 40%. On diagnosis, 53% of patients were taking nonsteroidal anti-inflammatory drugs. All patients were treated with second generation H1-antihistamines, but the standard dose was sufficient in only 18% of cases. Higher doses (up to 4 times the standard dose) achieved control of the urticaria in 74% of the patients studied. Higher doses of second generation H1-antihistamines were required to control the condition in patients with angioedema, and the presence of angioedema was associated with a lack of response to treatment with these drugs (OR, 6.1%; P<.001). One in 4 patients failed to respond to second generation H1-antihistamines and required treatment with omalizumab or ciclosporin to control their condition.
ConclusionsDoses of H1-antihistamines higher than the standard dose are required in most cases to achieve control of chronic spontaneous urticaria. Angioedema is associated with failure to respond to treatment with antihistamines. In refractory cases, control of the condition can be achieved with omalizumab or ciclosporin. Patients with chronic spontaneous urticaria do not generally avoid the use of nonsteroidal anti-inflammatory agents.
La urticaria crónica espontánea es una enfermedad prevalente, de difícil manejo terapéutico y con una importante repercusión en la calidad de vida del paciente.
ObjetivoDescribir las características epidemiológicas, clínicas y la respuesta terapéutica de los pacientes con urticaria crónica espontánea actuando según la guía de consenso EAACI/GA(2)LEN/EDF/WAO.
Material y métodoEstudio descriptivo transversal que incluyó todos los pacientes con urticaria crónica espontánea visitados en una consulta específica de alergia cutánea de un servicio de dermatología de un hospital terciario en España desde julio de 2011 hasta julio de 2015.
ResultadosCien pacientes con urticaria crónica espontánea participaron en el estudio. El 45% asociaba una urticaria inducible. El 40% asociaba angioedema. El 53% de los pacientes tomaba antiinflamatorios no esteroideos en el momento del diagnóstico. Todos los pacientes fueron tratados con antihistamínicos-H1 de segunda generación, pero solo un 18% se controló con dosis licenciadas, mientras que el aumento hasta 4 veces la dosis logró controlar el 74% de los pacientes. Los pacientes con angioedema requirieron dosis mayores de antihistamínicos-H1 de segunda generación que los pacientes sin angioedema para el control de la urticaria crónica espontánea, siendo el angioedema un factor asociado a falta de respuesta a tratamiento con antihistamínicos-H1 de segunda generación (OR 6,1%, p<0,001). Uno de cada 4 pacientes no respondió a antihistamínicos-H1 de segunda generación y requirió omalizumab o ciclosporina para su control.
ConclusionesEl control de la urticaria crónica espontánea precisa de la utilización de antihistamínicos-H1 de segunda generación a dosis más altas de las licenciadas en la mayoría de los casos. El angioedema se asocia a falta de respuesta al tratamiento con antihistamínicos. El omalizumab y la ciclosporina logran controlar los casos refractarios. La evitación de antiinflamatorios no esteroideos no es una práctica habitual en los pacientes con urticaria crónica espontánea.
Urticaria is one of the most frequent skin conditions in developed countries. It is characterized by recurrent and transient wheals and/or angioedema and can be classified according to its duration and cause.1,2 The wheals typically last less than 24hours. Angioedema involves the sudden onset of edema in the deep dermis and subcutaneous cellular tissue (which often affects the mucosa but may also affect other areas such as the face and extremities), can be accompanied by pain or itching, and resolves spontaneously in 72hours.2 The presence of wheals and/or angioedema with no known or inducible cause for more than 6 weeks is known as chronic spontaneous urticaria (CSU).2
CSU accounts for almost 70% of all cases of chronic urticaria and has a considerable effect on the patient's quality of life owing to its difficult therapeutic management.3 The 2009 international guidelines of the European Academy of Allergy and Clinical Immunology (EAACI), Global Allergy and Asthma European Network (GA2LEN), European Dermatology Forum (EDF), and World Allergy Organization (WAO) were aimed at harmonizing the definition, classification, diagnosis, and treatment of urticaria according to scientific evidence in the literature and taking into account experience with this condition in Europe and throughout the world.2,4 The guidelines were revised and updated in 2013 and are now the European consensus guidelines for management of CSU.5 According to the guidelines,4,5 if the condition does not respond to regular doses of second-generation H1 antihistamines (sgH1As), the recommended dose should be increased by as much as 4-fold. If this does not prove to be effective, the 2009 guidelines recommended switching the antihistamine as a third step in therapy. In refractory cases, the third line of treatment5 includes omalizumab, ciclosporin, or a leukotriene antagonist. The use of systemic corticosteroids would only be recommended for management of acute exacerbations in short regimens lasting less than 1 week.4,5
The primary objective of the present study was to describe the response to therapy of patients with CSU treated in the dermatology department of a tertiary hospital in Spain according to the consensus guidelines on therapy of EAACI/GA2LEN/EDF/WAO.4,5 The secondary objective was to describe the clinical and epidemiological characteristics of the patients studied.
Material and MethodsWe present a cross-sectional descriptive study of the epidemiologic and clinical characteristics and therapeutic management of patients with CSU followed at the skin allergy clinic of a dermatology department in a tertiary hospital in Spain from July 2011 to July 2015. The data were collected and analyzed in October 2015. We included all patients with idiopathic urticaria that had first appeared more than 6 weeks previously who were assessed in the clinic. We collected epidemiologic data (sex, age, previous episodes of urticaria, history of atopy, autoimmune diseases, and other relevant associated diseases), clinical data (months since onset of urticaria, date of onset of symptoms, date of diagnosis, types of associated urticaria, causes and aggravating factors, presence of angioedema, and adverse effects of treatment), and the results of laboratory tests (erythrocyte sedimentation rate, total serum immunoglobulin E, antinuclear antibodies, and antithyroid antibodies) and of skin prick testing. The treatment-related data collected were administration of oral corticosteroids (number of occasions, use in short regimens or continuous use for at least 1 month), use of antihistamines (type, dose at which urticaria is controlled), and use of antileukotrienes, ciclosporin, and omalizumab.
Treatment was considered to be effective when pruritus, wheals, and angioedema had disappeared or when the patient considered that the frequency and number of wheals and the relief from pruritus were sufficient and refused to start more aggressive treatments. Urticaria was considered to have resolved when the patient presented no new episodes of pruritus, wheals, or angioedema despite having stopped treatment for at least 3 months.
Data were collected from the electronic clinical history. A descriptive statistical analysis was performed, and factors associated with the lack of a response to sgH1As were evaluated using the chi-square test (IBM SPSS Statistics for Windows, Version 22.0, IBM Corp).
ResultsThe study population comprised 100 patients with CSU. Men accounted for 38% and women 62%. The mean age was 47.31 years. Mean time from onset of symptoms until diagnosis of CSU was 7.62 months. At the end of the study, urticaria had been resolved in 54% of cases with a mean (SD) time since onset of 23.20(18.72) months. A previous outbreak of urticaria was recorded in 17% of cases.
Types of associated urticariaOf the 100 patients with CSU, 40% also experienced angioedema.
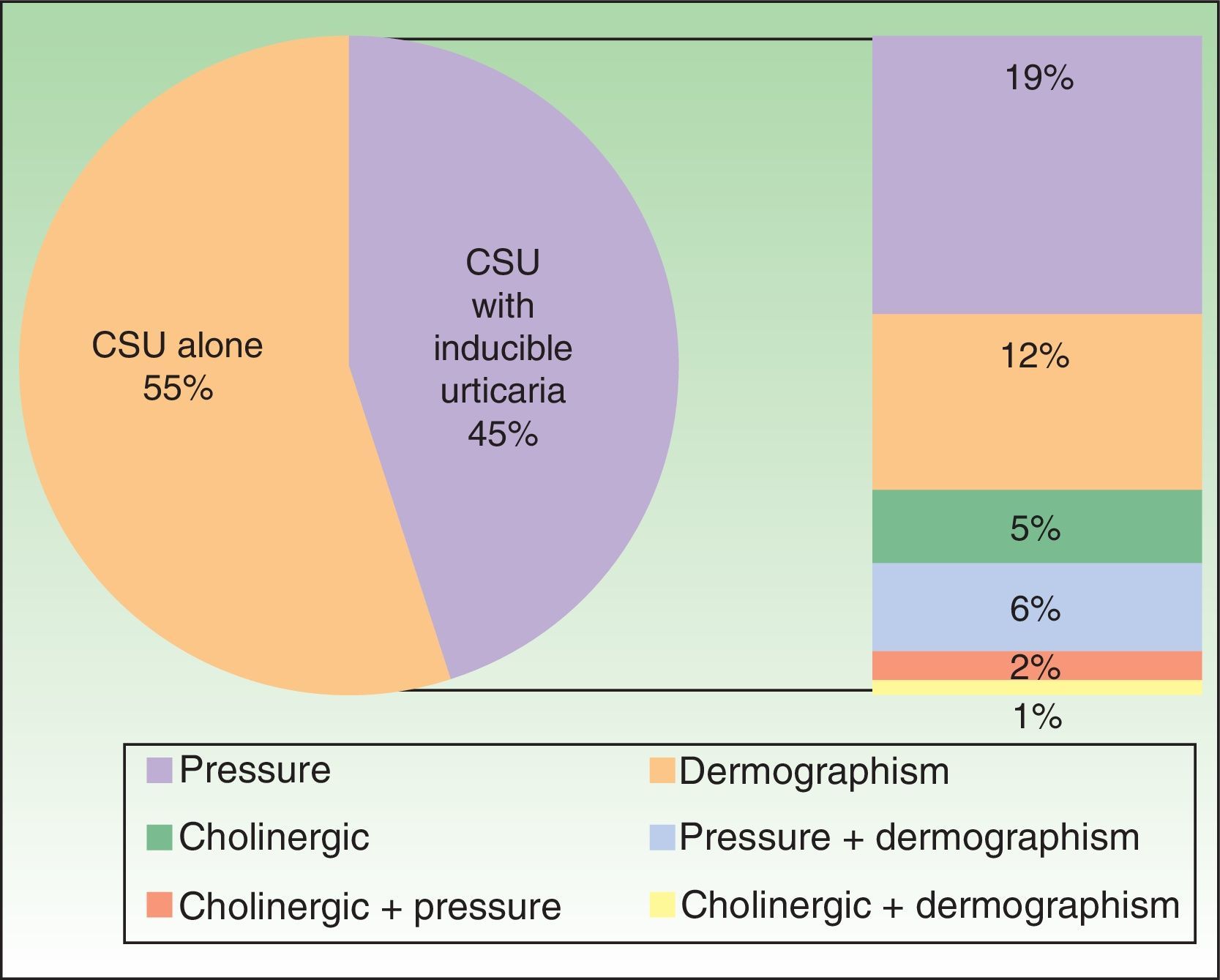
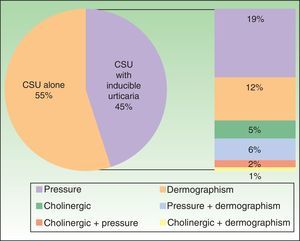
Furthermore, 45% of patients had some type of associated inducible urticaria (pressure, dermographism, and/or cholinergic) (Figure 1).
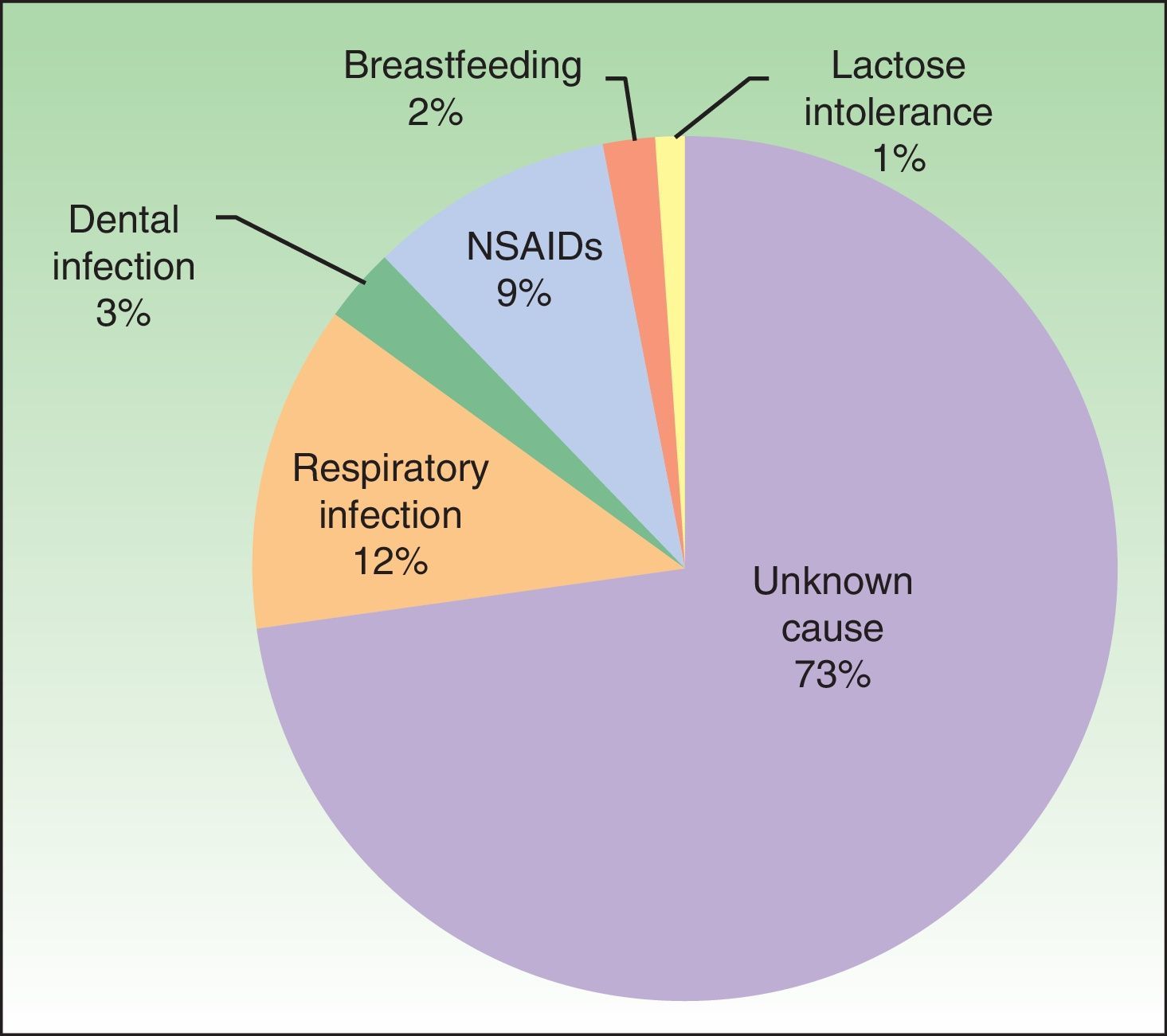
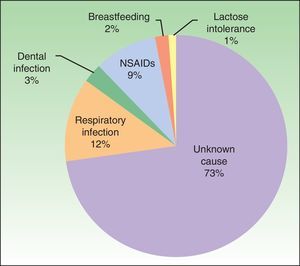
Possible causal agents and aggravating factorsAlthough all of the patients had idiopathic urticaria, 27% associated the onset of urticaria with a causal agent: infection, drugs (nonsteroidal anti-inflammatory agents [NSAIDs] in all cases), and other (Figure 2).
As for aggravating factors, 6 patients reported exacerbation of urticaria with NSAIDs. Taking into account these 6 patients and the 9 patients mentioned above implies that 15% of the total number of patients associated the onset or exacerbation of their disease with NSAIDs. In addition, 53% of patients were taking NSAIDs at the time of diagnosis.
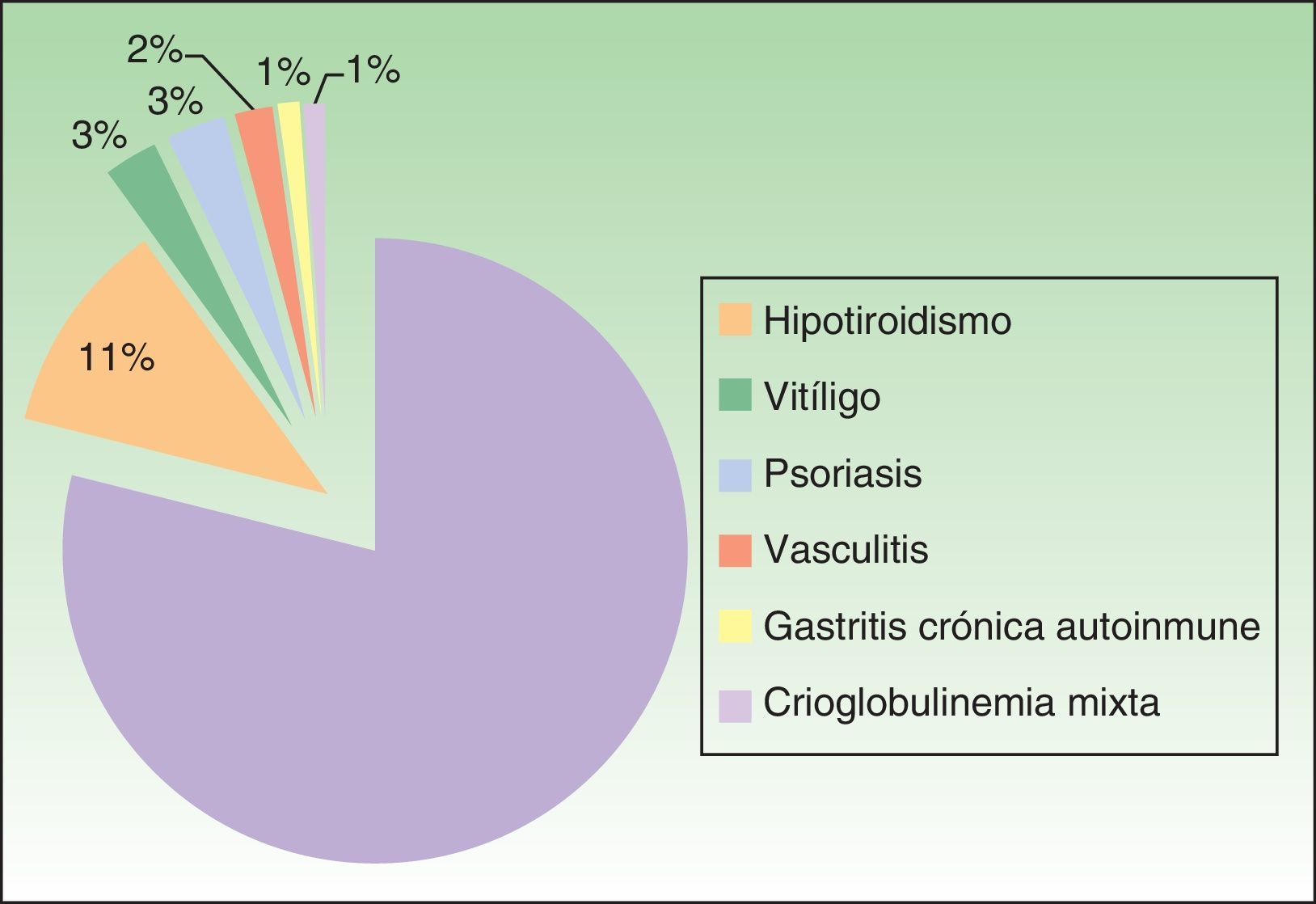
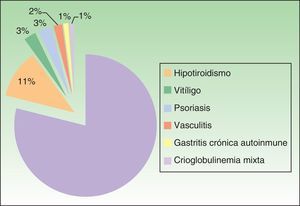
Associated diseases and additional testsOf the 100 patients with CSU, 4% had chronic infection by hepatitis B virus (HBV) or hepatitis C virus (HCV). In addition, 17% of patients had a personal history of atopy, and 20% had an autoimmune disease (Figure 3). The results of the laboratory tests showed that 36 of the 78 of patients tested for total serum IgE (46.1%) had levels that were higher than normal (>100IU/mL). Positive antinuclear antibody titers were detected in 10 of the 79 patients in whom this parameter was assessed (12.7%), and 20.3% of patients (14/69) had positive antithyroid antibody titers (and/or anti–thyroid-stimulating hormone antibodies). The result of the skin prick test was positive in 8 of the 40 patients who underwent it (20%), although relevance with respect to urticaria was unknown. The erythrocyte sedimentation rate was higher than normal (> 20mm/h) in 16 of 78 patients (20.5%).
Response to TreatmentSystemic corticosteroidsSystemic corticosteroids had to be administered at some point to control outbreaks in 56% of cases, on 2 occasions in 23.6% of cases, and on 3 or more occasions in 40% of cases. Treatment was mainly with oral deflazacort or prednisone at a dose that was tapered over 7-9 days.
Before evaluation in our unit, 9% of patients had received continuous corticosteroid therapy for at least 1 month.
AntihistaminesAll patients started sgH1As as their first-line treatment. Before switching to another treatment, 60% of patients took 2 or more different sgH1As and 26% took 3 or more. Before attending our clinic, 43% of patients had received sedating H1 antihistamines.
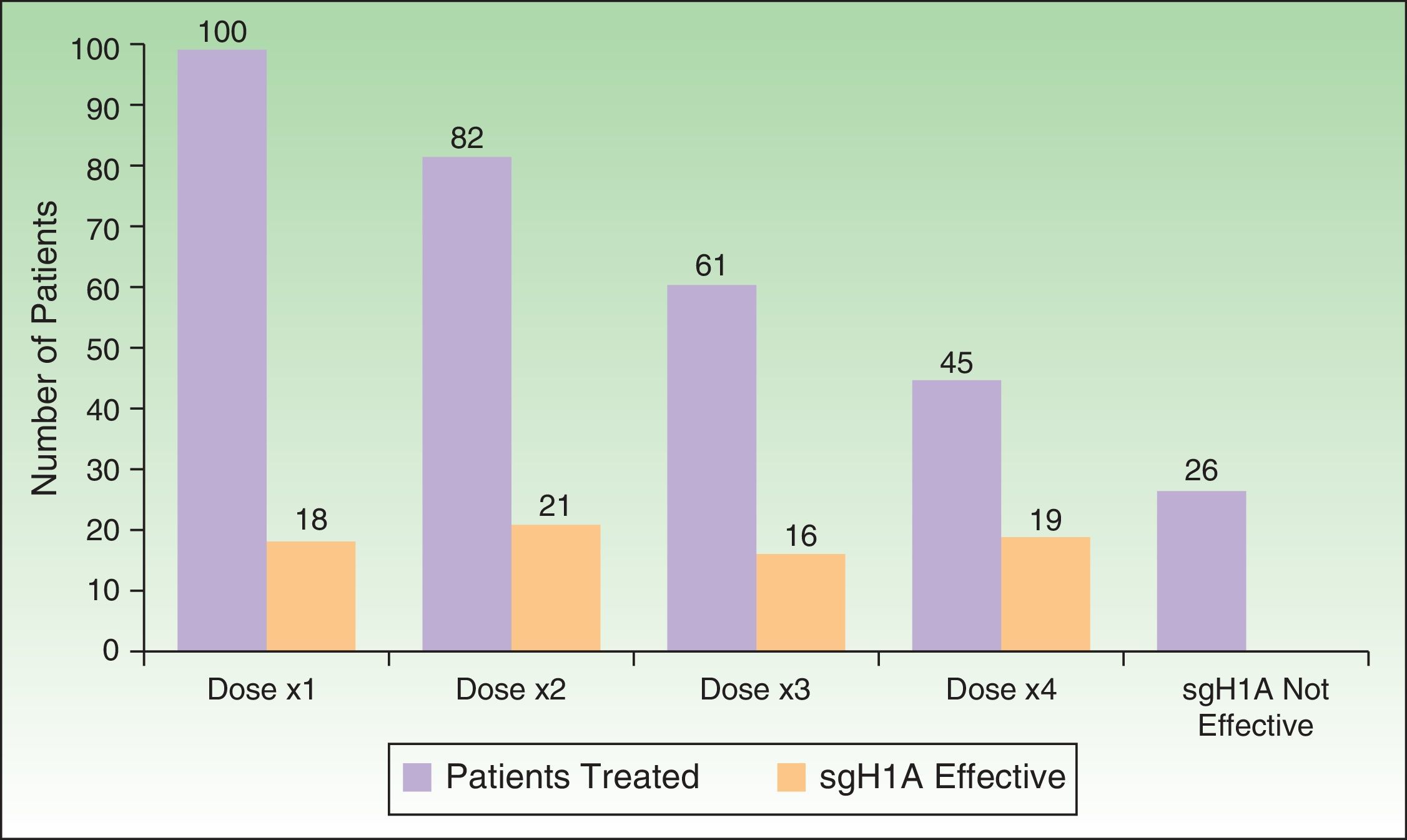
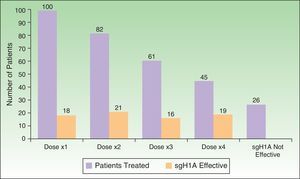
As for the response to treatment (Figure 4), the disease was controlled with a single daily dose of sgH1A in 18% of cases, whereas it was necessary to double, triple, or quadruple the dose in 82%. The condition remained uncontrolled with sgH1As in 1 of every 4 patients (26%), thus necessitating other lines of treatment.
AntileukotrienesThe leukotriene receptor antagonist montelukast was administered at 10mg/d to 34% of patients together with triple- or quadruple-dose antihistamine treatment. Montelukast proved to be effective in 1 of every 3 patients treated (38.2%).
CiclosporinA total of 11 patients (11%) were treated with ciclosporin as the third or fourth line of treatment at doses ranging from 3.5 to 4mg/kg/d. This approach was effective in 63.6% of cases.
No patients had to suspend treatment because of adverse effects or developed severe adverse effects at the dose administered. All of the patients combined treatment with sgH1As.
OmalizumabA total of 16 patients (16%) were treated with omalizumab. Of these, 12 (75%) received the drug as their third line of treatment and 4 (25%) as their fourth line of treatment after the failure of ciclosporin. Omalizumab proved effective for the control of urticaria in all cases.
The doses administered ranged from 150mg/mo to 375mg every 15 days. None of the patients experienced adverse effects to treatment. Treatment was successful in 2 patients with active HCV infection and 1 patient with active HBV infection. Previous treatment with montelukast±sgH1A was suspended in 44% of patients taking omalizumab owing to the efficacy of omalizumab in monotherapy.
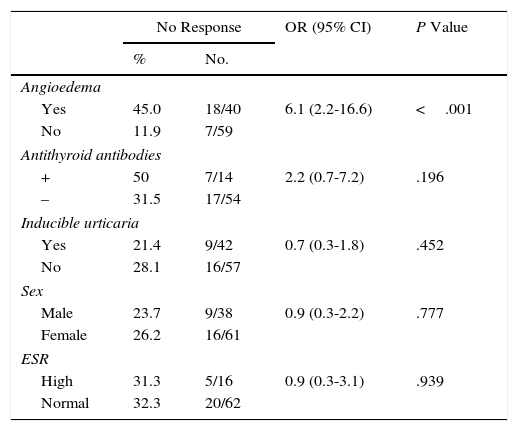
Factors associated with the lack of response to treatment with sgH1AsThe factors analyzed as being potentially associated with lack of response to treatment with sgH1As were as follows: sex, angioedema, inducible urticaria, antithyroid antibodies, and high erythrocyte sedimentation rate (Table 1). Angioedema was associated with a greater probability of not responding to sgH1As (OR, 6.1; 95% CI, 2.2-16.6; P<.001), with an up to 6-fold greater risk than patients who did not experience angioedema. Patients with positive antithyroid antibody titers showed a greater tendency not to respond to sgH1As than patients with negative antibody titers (OR, 2.2; 95% CI, 0.7-7.2; P=.196). No relevant or statistically significant differences were found for the remaining factors analyzed.
Frequency and Factors Associated With Lack of Response to Second-Generation H1 Antihistamines.
| No Response | OR (95% CI) | P Value | ||
|---|---|---|---|---|
| % | No. | |||
| Angioedema | ||||
| Yes | 45.0 | 18/40 | 6.1 (2.2-16.6) | <.001 |
| No | 11.9 | 7/59 | ||
| Antithyroid antibodies | ||||
| + | 50 | 7/14 | 2.2 (0.7-7.2) | .196 |
| – | 31.5 | 17/54 | ||
| Inducible urticaria | ||||
| Yes | 21.4 | 9/42 | 0.7 (0.3-1.8) | .452 |
| No | 28.1 | 16/57 | ||
| Sex | ||||
| Male | 23.7 | 9/38 | 0.9 (0.3-2.2) | .777 |
| Female | 26.2 | 16/61 | ||
| ESR | ||||
| High | 31.3 | 5/16 | 0.9 (0.3-3.1) | .939 |
| Normal | 32.3 | 20/62 | ||
Abbreviations: CI, confidence interval; ESR, erythrocyte sedimentation rate; OR, odds ratio; sgH1A, second-generation H1 antihistamines.
Ours is the first study in Spain to investigate management of patients with CSU in daily clinical practice based on the indications of the most recently published consensus guidelines.2,4,5 The epidemiological data in our study are consistent with those in the literature, namely, that CSU mainly affects middle-aged women.6–8 Data on the association with angioedema (40%) are also consistent with those reported in the literature.6,8 However, it is noteworthy that the association with inducible urticaria was more common in our study (45%) than in other studies,8,9 possibly as a reflection of the effort we have been making to detect inducible urticaria based on a meticulous clinical history and challenge tests (eg, scraping and Fric Test, Temp Test, hot water immersion test, exposure to sunlight, and physical exercise).10 Knowing that the patient is affected by inducible urticaria can help us to prevent the factors that trigger this condition. In addition, we believe that test results play a key role in the management of the disease, since some authors suggest that the response to treatment is worse in patients with CSU and angioedema and/or physical or inducible urticaria,8,9 as we observed in patients with angioedema in our study, who had a 6-fold greater risk of not responding to sgH1As than patients who did not have angioedema (P<.001).
The causes and aggravating factors associated with CSU are diverse and vary depending on the study. We found that 27% of patients associated onset of CSU with a causal agent. This finding is consistent with data from the literature,11 although the percentage is lower than expected according to a recent study in which the authors suggest that a more intensive diagnostic workup in tertiary centers can reveal a higher number of patients for whom the cause of CSU is identified.12 Infection and medication, as well as chronic diseases such as hepatitis, are recognized as the main causes to be identified in the EAACI/GA2LEN/EDF/WAO guidelines,2 other published studies,13 and our study. We found that 4 patients (4%) had chronic HCV or HBV infection; this percentage is much higher than the prevalence in the general population in Spain, which, according to published data, ranges from 0.27% to 2.6%.14 The close association between CSU and NSAIDs detected in our study (15%) is significantly higher than reported in the literature.15 We know that drugs such as NSAIDs, codeine, and opiate derivatives can induce or exacerbate chronic urticaria through direct action on mast cell degranulation.16 The EAACI/GA2LEN/EDF/WAO guidelines2 recommend that the diagnostic workup for CSU routinely include blood tests with a complete blood count and C-reactive protein and the exclusion of suspect drugs such as NSAIDs. This observation is particularly relevant in the diagnostic and therapeutic workup of patients with CSU in daily practice, since up to 53% of patients in our study were taking NSAIDs at the time of their diagnosis. NSAIDs are widely used in Spain, generally to treat specific episodes and often without prescription. We believe that this drug family should be avoided in patients with CSU and we should make every effort to provide appropriate education in this area for both patients and health professionals.
As for the other tests we performed during the study, the prevalence of high serum IgE levels and positive prick test results is well known in patients with CSU, even though IgE-mediated chronic urticaria is exceptional.17 Autoreactive or autoimmune chronic urticaria is one of the most frequent subtypes of CSU7 and is diagnosed using the autologous serum skin test and basophil degranulation test, with a prevalence of positive results of up to 67% in some studies.7,18 One of the limitations of our study is that we cannot provide the percentage of cases of autoreactive CSU, since we do not have the equipment necessary to perform the autologous serum skin test. Nevertheless, it is worth mentioning that 41% of patients had indirect signs of autoreactivity (positive antinuclear antibody titers and/or positive antithyroid antibody titers and/or associated autoimmune disease). A positive result in the autologous serum skin test is associated with greater resistance to treatment, more intense activity and duration of urticaria, and worse quality of life.7,8 In our study, we observed a trend towards a poorer response to treatment with sgH1As in patients with positive antithyroid antibodies than in patients without indirect signs of autoreactivity, although the differences were not statistically significant. The EAACI/GA2LEN/EDF/WAO guidelines recommend performing only a complete blood count and C-reactive protein as routine practice.2 However, we recommend that the basic workup for patients with CSU also include a thyroid profile and antithyroid antibody test.
The response to treatment in the present study was as expected. Treatment with sgH1A at the recommended doses controlled the condition in a small percentage of patients with CSU (18%); most required their dose to be increased. Administration of almost 4-fold the recommended dose of sgH1A controlled the condition in 3 of every 4 cases. Nevertheless, 26% of patients did not respond to sgH1As and required other lines of treatment. These data are consistent with those published elsewhere,8,19–21 which show the efficacy and safety of quadrupling the dose of sgH1A as second-line therapy, as recommended in the guidelines.4,5 It is noteworthy that more than one-third of the patients in the present study (43%) had been treated with sedating H1 antihistamines before attending our clinic. This treatment is not recommended for management of CSU in guidelines4,5 or scientific studies,22,23 although it is considered by many to be safe owing to the fact that it has been in use for a long time and is commonly added before bedtime because it is thought to reduce itching and improve sleep quality.22 Nevertheless, it has been shown to diminish academic performance and work productivity, and it has been associated with traffic accidents and death by overdose.23 In line with other studies,11 we observed that many patients receive sedating H1 antihistamines together with oral corticosteroids as first-line therapy when they attend medical centers where the specialists are not acquainted with the guidelines or that they self-medicate owing to the fact that the drugs can be acquired without prescription. We believe that it could be extremely interesting to update management of CSU for primary care specialists, since, in most cases, these are the first physicians to evaluate and treat the condition.
The update of the international guidelines5 reduced the therapeutic algorithm to 3 steps, placing leukotriene receptor antagonists, ciclosporin, and omalizumab at the same level on the last step. In our study, montelukast was effective in 1 of every 4 patients treated. We believe that montelukast is safe and easy to use and that it can prove effective on occasion.
Ciclosporin was effective in most treated patients. It proved to be safe and well tolerated at 3.5-4mg/kg/d. The results of published studies24,25 are consistent with ours in terms of efficacy and safety, although the doses are slightly lower (3mg/kg/d).
Several published studies have examined the administration of omalizumab for the treatment of CSU in daily clinical practice.26–30 In our study, omalizumab made it possible to control cases that were refractory to almost all of the treatments in the guidelines,4,5 with 100% efficacy and no adverse effects. Long-term studies are necessary to enable us to adjust the indications, doses, and maintenance period, as well as to determine long-term safety and when and how to discontinue the drug.31 However, in our experience, the drug brings hope for cases of CSU that are refractory to conventional treatment options.
LimitationsThe main limitations of the present study are the lack of measures for the clinical evaluation of the activity of urticaria and quality of life and the fact that we were unable to evaluate CSU using the autologous serum skin test, which was not available at our center.
We currently monitor urticaria and evaluate its impact on quality of life using validated tools, namely, the Urticaria Activity Score, which enables us to evaluate the activity of urticaria and response to treatment, and the Urticaria Control Test and the Angioedema Activity Score.
Current recommendations for adequate management of patients with CSU in daily practice include, at least, the Urticaria Activity Score and the Urticaria Control Test.
ConclusionsCSU is a complex disease that requires a holistic approach to the patient. An exhaustive clinical history should be taken to rule out the main causes and triggers. Symptoms should be treated as soon as possible. Use of NSAIDs by patients with CSU continues to be common, and we should educate both patients and health professionals to avoid these drugs or replace them with other analgesics. Angioedema is very common in CSU. It is therefore important to question patients carefully and recognize its presence, since it is associated with a lack of response to sgH1As.
Administration of sgH1As at recommended doses only controls CSU in 1 out of every 5 patients. However, increasing the dose by up to 4-fold makes it possible to control the condition in 74% of patients. Every effort should be undertaken to make physicians more aware of this approach so that they can take appropriate measures.
Ciclosporin and omalizumab are effective and safe for control of CSU that is refractory to first-line options. We believe that patients with CSU should be managed in reference units.
By following European guidelines, we were able to control CSU in almost all of the patients studied. Therefore, we encourage all dermatologists to apply the guidelines in their clinical practice.
Ethical DisclosuresProtection of humans and animalsThe authors declare that no tests were carried out in humans or animals for the purposes of this study.
Confidentiality of dataThe authors declare that they have followed their institutional protocols on publication of patient data.
Right to privacy and informed consentThe authors declare that no private patient data appear in this article.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Marín-Cabañas I, Berbegal-de Gracia L, de León-Marrero F, Hispán P, Silvestre JF. Manejo de la urticaria crónica espontánea en la práctica clínica diaria siguiendo las indicaciones de la guía EAACI/GA(2)LEN/EDF/WAO. Actas Dermosifiliogr. 2017;108:346–353.