Although the diversity of the clinical manifestations of syphilis is well-known, atypical presentations can also occur. Such atypical presentations are associated with a high risk of transmission as a result of diagnostic confusion and treatment delays owing to the disease's ability to mimic other common skin diseases, deviate from classic clinical presentations, and adopt unique forms. Cases of atypical syphilis have been described most frequently in patients with concomitant human immunodeficiency virus (HIV) infection. Because the incidence of syphilis has been growing over recent years —particularly in patients with HIV co-infection—dermatologists need to be familiar with the less well-known clinical presentations of this venereal disease.
Aunque es por todos bien conocida la heterogeneidad de manifestaciones clínicas con la que puede presentarse la sífilis, esta también puede mostrarse con patrones atípicos.
Ya sea por su capacidad de mimetizar otras dermatosis conocidas, por saltarse los patrones clínicos clásicos o por su originalidad clínica, la sífilis en sus formas atípicas puede llevar a confusiones en el diagnóstico, retrasos en el tratamiento y por tanto un alto riesgo de transmisión.
Entre pacientes coinfectados por el VIH se han descrito más frecuentemente casos de sífilis atípica. El hecho de que se trate de una infección venérea cuya incidencia está aumentando en los últimos años, especialmente en pacientes con VIH, hace necesario que los dermatólogos se familiaricen también con estas formas clínicas menos conocidas.
Syphilis has been very appropriately named the great imitator and is of considerable clinical interest due to its wide diversity of clinical—and dermatologic—manifestations. The resurgence of syphilis in recent years has required newer generations of dermatologists to become familiar with the clinical variants of this disease, and particularly those that deviate from classical presentations.
The organism responsible for syphilis, Treponema pallidum, penetrates its host through intact mucous membranes and/or skin wounds and then multiples and spreads through the body. Thirty percent of sexual partners of recently infected patients develop syphilis.
Syphilis continues to be a worldwide problem,1 and in many countries, including Spain, it is a notifiable disease.
The annual incidence of syphilis has risen steadily in the United States since 2001, with data from 2013 indicating a rate of 5.3 cases of early syphilis per 100000 inhabitants.2 There has also been a notable increase in new cases in Spain, with rates doubling between 1995 and 2010.3
According to numerous reports, the greatest increase in the incidence of syphilis has occurred in men who have sex with men (MSM), largely due to their engagement in risky sexual practices (e.g., sex with unknown partners, unprotected sex, promiscuity, and use of illicit drugs, in particular, methamphetamines).4–10
Concomitant syphilis and human immunodeficiency virus (HIV) infection is very common, as the 2 conditions have the same route of infection. In addition, the presence of one infection facilitates the acquisition and transmission of the other.11,12 Discharge from syphilis lesions is rich in T cells and dendritic cells. The fact that many of these cells express HIV coreceptors (CCR5 and DC-SIGN) could explain the epidemiological relationship between the 2 pathogens.
The estimated rate of concomitant primary or secondary syphilis and HIV infection in MSM in the United States is 86%.13,14 The prevalence of syphilis and HIV coinfection in MSM in Spain, however, is estimated to be much lower, with data from recent years indicating a rate of around 28%.15
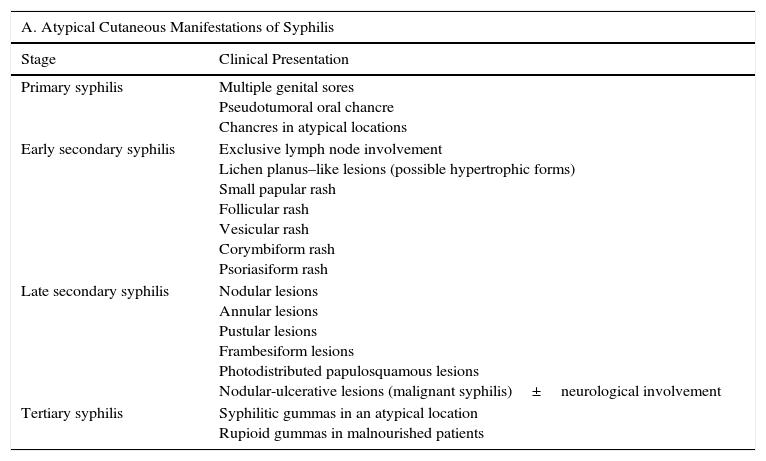
Atypical Clinical ManifestationsIn the absence of appropriate treatment for T pallidum infection, the disease progresses through different stages, each of which has characteristic clinical manifestations. Nonetheless, reports of uncharacteristic, i.e., atypical, manifestations have existed for years (Table 1). Syphilis also has other clinical manifestations that, while well-characterized and considered typical, are uncommon. In this review, we focus on atypical manifestations.
Atypical Cutaneous Manifestations of Syphilis in Seronegative Patients (A) and in Patients with Human Immunodeficiency Virus Infection (B).
| A. Atypical Cutaneous Manifestations of Syphilis | |
|---|---|
| Stage | Clinical Presentation |
| Primary syphilis | Multiple genital sores Pseudotumoral oral chancre Chancres in atypical locations |
| Early secondary syphilis | Exclusive lymph node involvement Lichen planus–like lesions (possible hypertrophic forms) Small papular rash Follicular rash Vesicular rash Corymbiform rash Psoriasiform rash |
| Late secondary syphilis | Nodular lesions Annular lesions Pustular lesions Frambesiform lesions Photodistributed papulosquamous lesions Nodular-ulcerative lesions (malignant syphilis)±neurological involvement |
| Tertiary syphilis | Syphilitic gummas in an atypical location Rupioid gummas in malnourished patients |
| B. Atypical Manifestations of Syphilis in Patients With Human Immunodeficiency Virus Infection | |
|---|---|
| Primary syphilis | Overlapping of stages Multiple genital chancres Pseudotumoral oral chancre Early-stage neurosyphilis Syphilitic gummas in early infection stages |
| Secondary syphilis | Overlapping of stages Atypical cutaneous manifestations of secondary syphilis described in Section A Mycosis fungoides–like lesions Early-stage neurosyphilis Syphilitic gummas in early infection stages |
| Tertiary syphilis | Overlapping of stages Syphilitic gummas in an atypical location Syphilitic aortitis with rapid progression Asymptomatic neurosyphilis Otosyphilis Ocular syphilis |
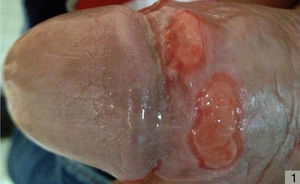
About 2 to 3 weeks after infection by T pallidum, a painless papule generally develops at the site of inoculation; this then grows and ulcerates, leaving a hard, round, ulcerative lesion known as a chancre, that is not painful. Chancres are generally solitary, and the presence of multiple sores in a patient with primary syphilis can be considered an atypical manifestation (Fig. 1).
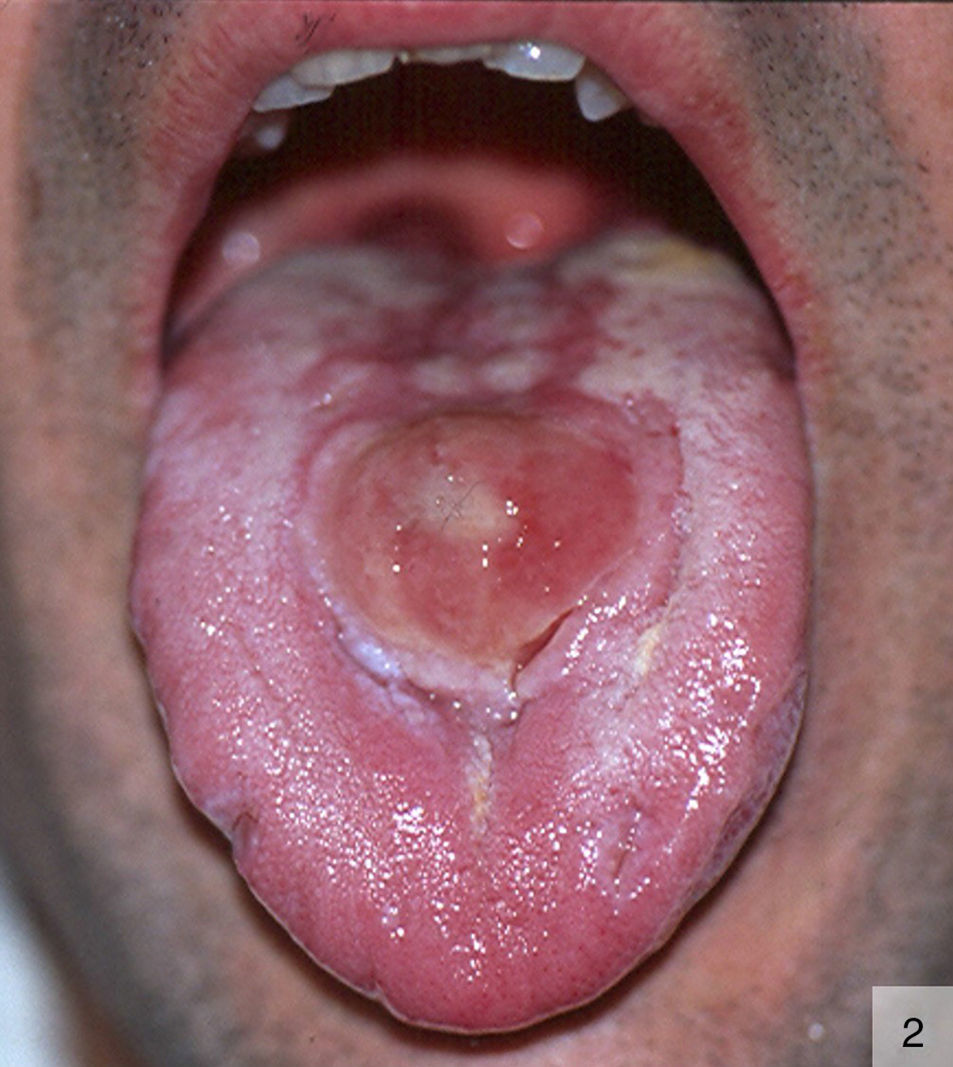
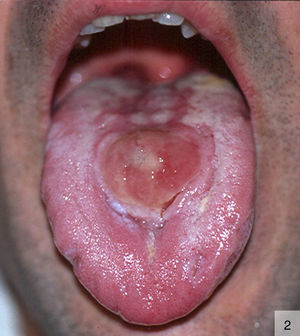
Other atypical variants of primary syphilis reported include pseudotumoral chancres on the dorsum of the tongue16 (Fig. 2) and, more recently, a primary chancre on the hand associated with regional lymphadenopathy.17
Secondary SyphilisIf left untreated, primary syphilis progresses to secondary syphilis in the vast majority of patients. This is the most florid stage of disease and is characterized by the spread of organisms to the tissues. It can be accompanied by nonspecific symptoms (e.g., fever, malaise, headache, sore throat) in 12% of patients.
Other uncommon but typical manifestations of secondary syphilis are iritis, anterior uveitis, osteitis and periostitis,18 glomerulonephritis, hepatitis, and nephrotic syndrome.19–23
Generalized lymphadenopathy, without pain, accompanies manifestations of secondary syphilis in 70% to 85% of patients.24 In 1 report, a patient had exclusive lymph node involvement, leading to an initial suspicion of lymphoma, but polymerase chain reaction subsequently identified treponemes in the lymph nodes.25
Lichen planus–type lesions, which tend to be nonpruritic, have also been described as an atypical presentation of secondary syphilis. In 1 recent report, treponemes were identified by immunohistochemistry in a patient with hypertrophic lichen planus.26
Other atypical presentations of early secondary syphilis are small papular, follicular, vesicular, corymbiform, and psoriasiform rashes. Corymbose syphilis, whose name comes from the Greek term kórymbos, meaning top, or convex cluster of fruit or flowers, presents clinically as a central plaque surrounded by a ring of discrete papules. This atypical form has been described in a woman reinfected with syphilis.27
Nodular, annular, pustular, frambesiform, nodular-ulcerative (malignant) forms of late secondary syphilis can also be easily confused with other skin conditions (Table 2).28
Atypical Cutaneous Manifestations of Secondary Syphilis and Differential Diagnosis.
| Nodular syphilis | Lymphoproliferative diseases, deep fungal infections, lepromatous leprosy, tuberculosis, sarcoidosis, foreign body reactions, adnexal tumors, granular cell tumors, scleromyxedema, lipoid proteinosis, lymphoma, leukemia cutis, histiocytosis |
| Annular syphilis | Lichen planus, subacute cutaneous lupus erythematosus, sarcoidosis, atypical mycobacterial infection, granuloma annulare, erythema annulare centrifugum, scabies (in cases of male genital involvement) |
| Pustular syphilis | Folliculitis, boils, papulopustular rosacea, ecthyma, primary pyoderma gangrenosum lesion, steroid acne, Behçet disease, Sweet syndrome |
| Frambesiform syphilis | Common warts, pinta, endemic syphilis |
| Malignant syphilis | Ecthyma, ecthyma gangrenosum, anthrax, blastomycosis-like pyoderma, pyoderma gangrenosum, atypical mycobacterial infection, deep fungal infection, vasculitis |
| Photodistributed papulosquamous rash | Systemic lupus erythematosus, dermatomyositis, subacute lupus erythematosus, seborrheic dermatitis, rosacea, polymorphous light eruption, photo-induced lichenoid reaction |
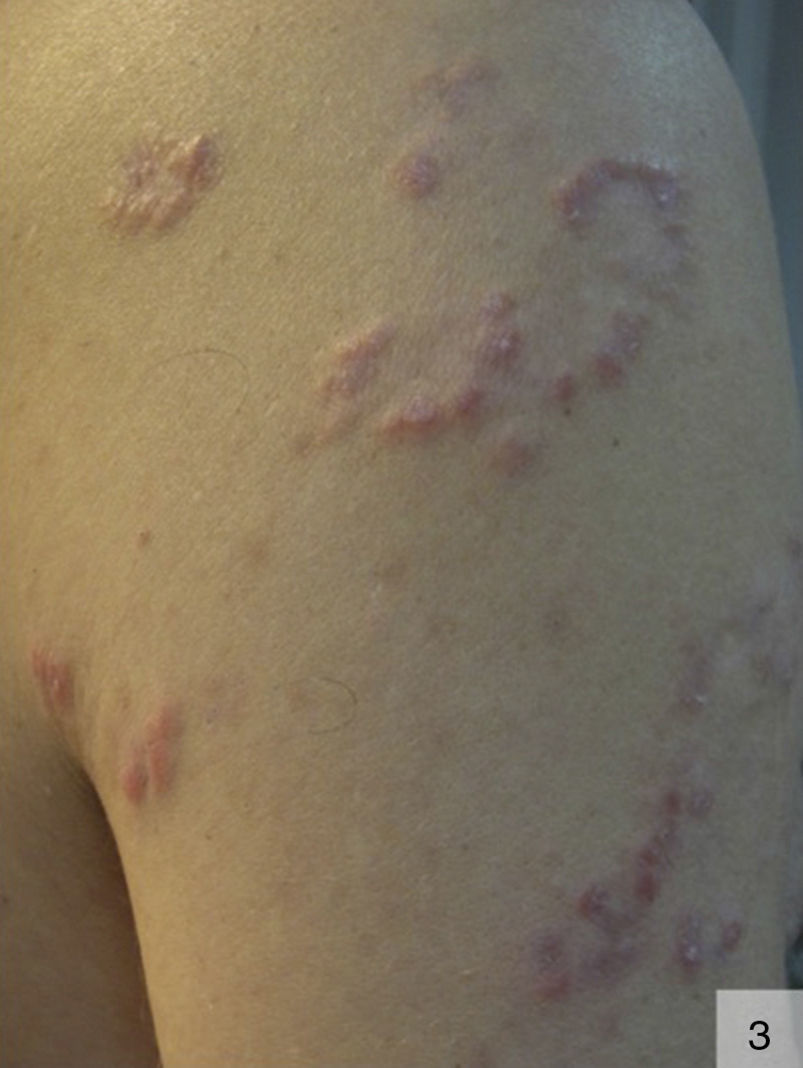
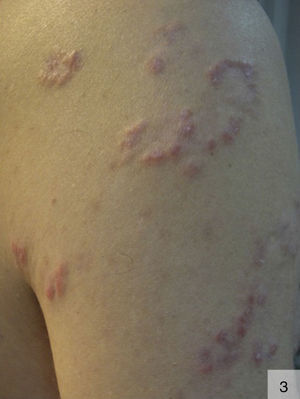
The nodular variant of secondary syphilis can present as erythematous or violaceous plaques or nodules that can form a localized or generalized symmetric rash. These lesions can occasionally adopt an arciform pattern (Fig. 3).29 The palms, soles, and mucosal surfaces tend to be spared.30 Solitary lesions have also been described on the scalp, the dorsum of the tongue, the lip, and the areola mammae. The histologic features of nodular syphilis are variable, and can include acanthosis and a perivascular and periadnexal lymphoplasmacytic infiltrate.31 Other features include lichenoid infiltrates with vacuolar degeneration of the basement membrane. Histologic features similar to those seen in pseudolymphoma and follicular lymphoma have also been described.32 Nodular secondary syphilis tends to be associated with nonnecrotizing granulomas, which can be difficult to differentiate from lesions seen in sarcoidosis but in which treponemal epitopes may be identified.33
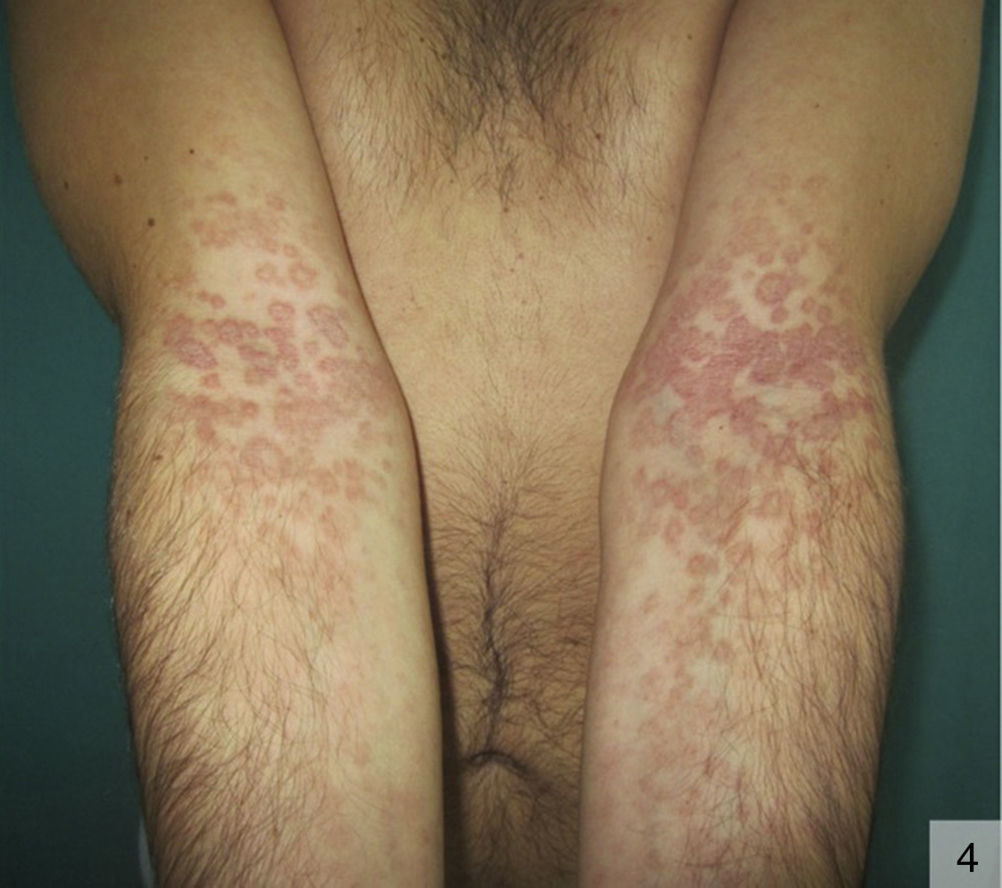
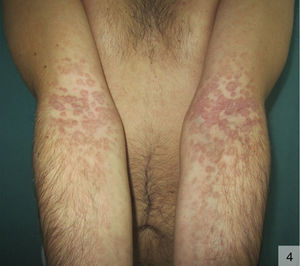
The morphology of annular secondary syphilis is highly variable (Fig. 4), with lesions ranging from fine, slightly scaling papules to exophytic verrucous forms that can affect the scalp, the trunk, and the perioral, perianal, and genital regions. Scalp involvement can cause scarring alopecia. Typical locations are oral commissures, the scalp, palms and soles, and intertriginous areas.34
There has also been a report of a variant of syphilis with concentric, delicate, slightly scaling lesions mimicking tinea imbricata.35 Histology may show a lichenoid or focal perifollicular infiltrate of plasma cells. Additional features can include interphase dermatitis, plasma cells, and a predominant lymphoplasmacytic perivascular infiltrate. In patients with verrucous lesions on clinical examination, histology may show epidermal hyperplasia, irregular acanthosis, spongiosis, and parakeratosis.36 Granulomatous reactions with giant multinucleated, epithelioid cells, similar to those seen in nodular secondary syphilis, may also be observed.34
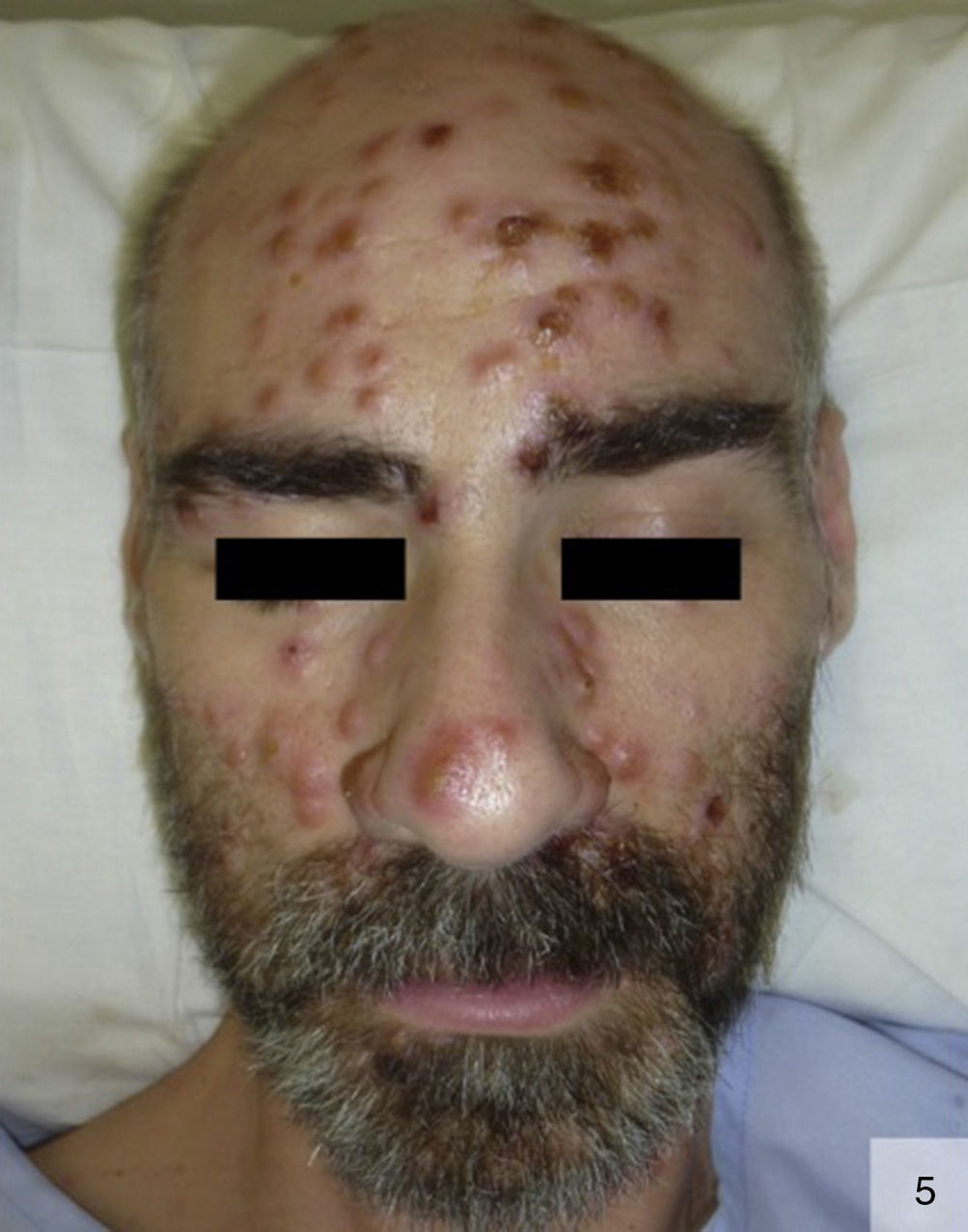
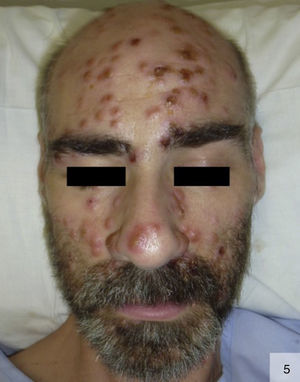
There have been few reports of pustular secondary syphilis in the literature (Fig. 5).37–39 Primary lesions are surrounded by follicles and are symmetrically distributed on the face, trunk, and extremities, with possible involvement of the palms and soles. These lesions form a crust, and can frequently leave scars or postinflammatory pigmentary changes.38 Histology can show collections of intrafollicular or subcorneal neutrophils, together with focal epidermal necrosis and perivascular lymphoplasmacytic infiltrates.37 Noncaseating granulomas have also been described.37 Histology, however, may only reveal spongiosis with exocytosis, dermal edema, and a perivascular lymphocytic infiltrate.
Frambesiform secondary syphilis refers to vegetating, ulcerative, keratotic lesions, which are at times moist and tend to appear in periorificial regions and on the trunk and extremities, although they may be limited to the scalp. This form of secondary syphilis is very uncommon.40,41 Although there may be a pustular component, in this case, unlike in pustular secondary syphilis, the pustules are not centered around the follicles. Histology shows epidermal acanthosis with neutrophilic infiltration and a prominent plasma cell infiltrate.42
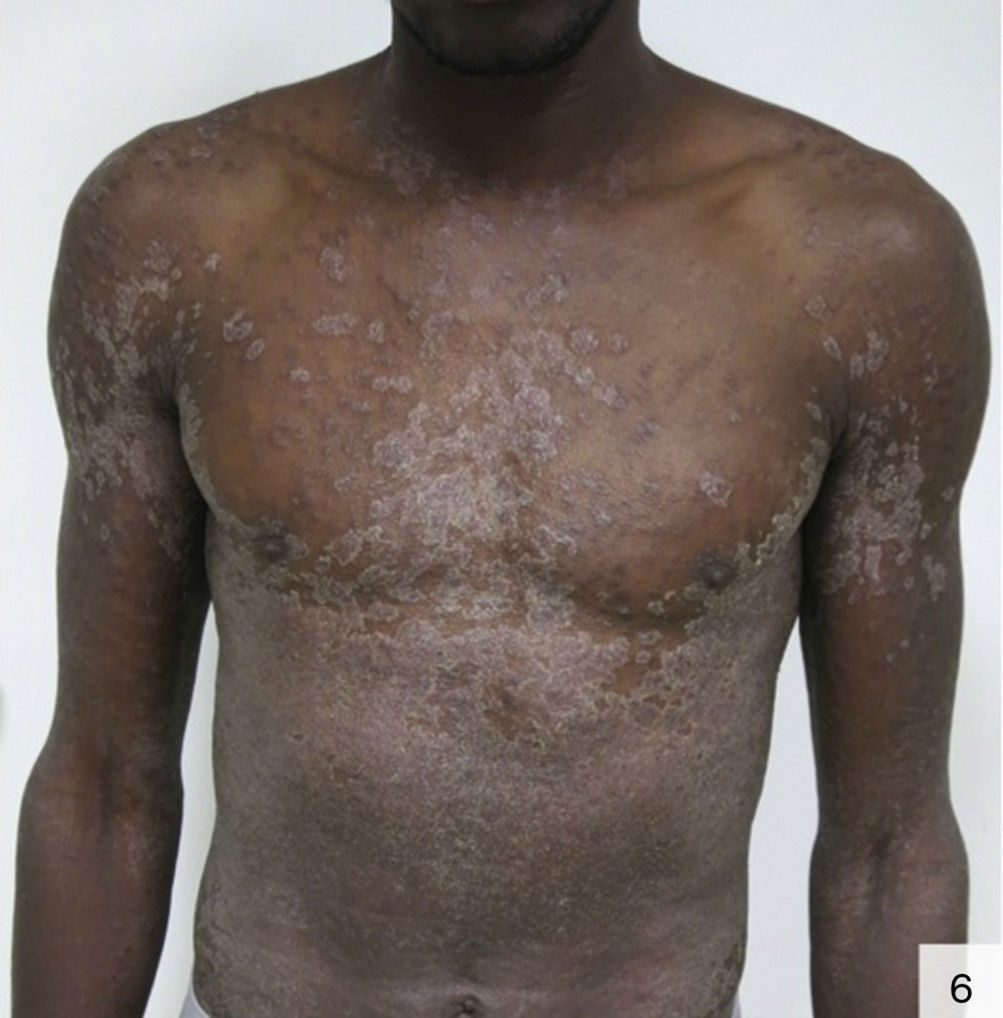
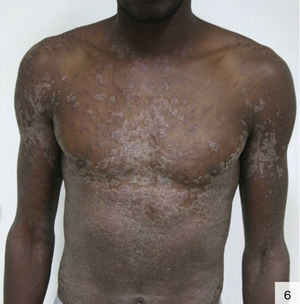
There has also been a report of photodistributed papulosquamous syphilis that can give the skin an ichthyosiform appearance. Diagnosis of the above forms of syphilis requires a high index of suspicion (Fig. 6).
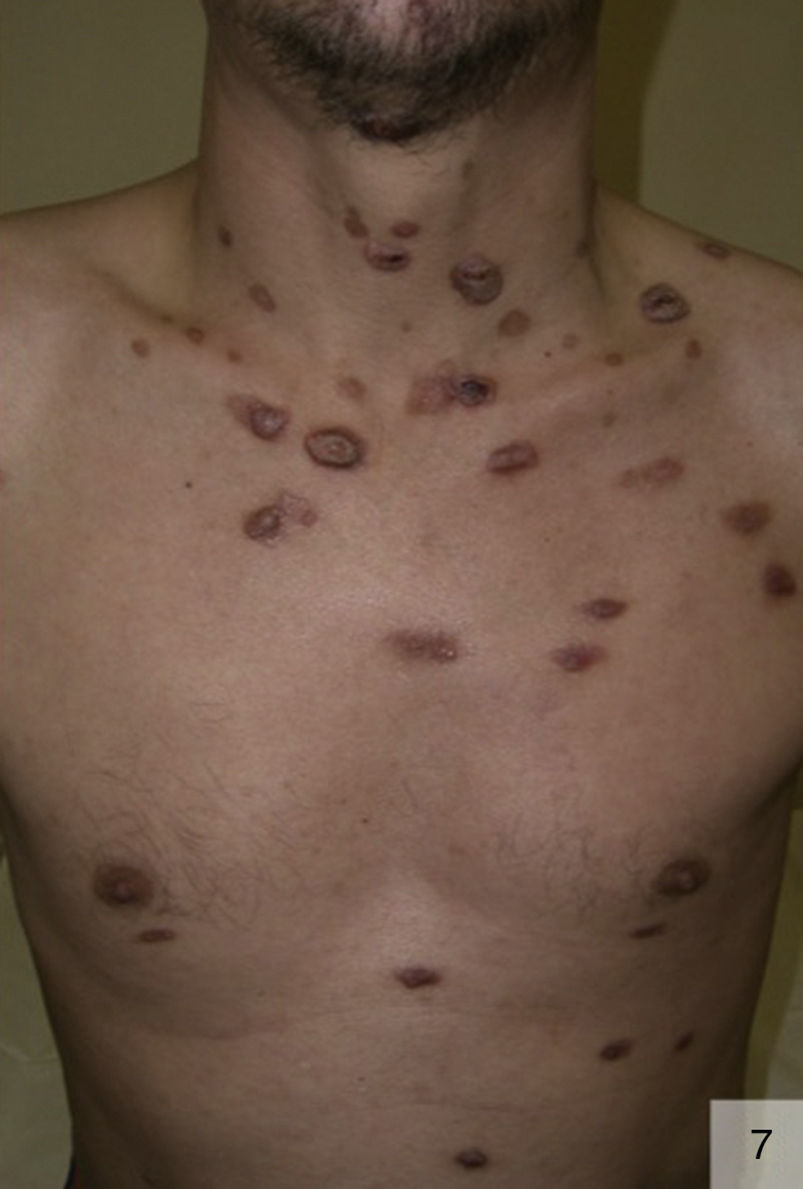
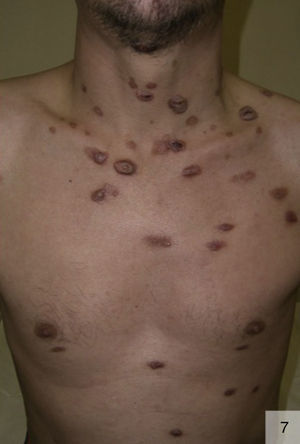
Malignant syphilis, also called nodular-ulcerative syphilis (Fig. 7) or leus maligna, is a rare but severe form of secondary syphilis that has been described largely in MSM with HIV coinfection, although it can also occur in immunocompetent individuals and heterosexuals who engage in risky sexual practices.43 Malignant syphilis is characterized by markedly prodromal symptoms (fever, chills, headache, general malaise, weight loss, and generalized lymphadenopathy), followed by disseminated, irregularly distributed lesions that are clinically similar to primary chancres. The lesions start off as papules and then progress to oval, well-delimited ulceronecrotic plaques. There may also be lesions at different clinical stages, in addition to mucosal involvement. Malignant secondary syphilis can present with neurological symptoms and spirochetes may be isolated in cerebrospinal fluid (CSF).44 Approximately 50% of patients with malignant syphilis have CSF abnormalities, although the majority do not have neurological manifestations.45
One recent report described a case of Bell's palsy in an immunocompetent patient with malignant syphilis.46 There has also been a case of ocular involvement in a seropositive homosexual man with malignant syphilis in whom it was not possible to isolate T pallidum by either PCR or immunohistochemical staining.47 Analysis of patients with active syphilis has shown that malignant syphilis is 60 times more common in those with HIV coinfection.48
The use of immunohistochemical staining to identify T pallidum in cutaneous lesions of malignant syphilis has become commonplace in recent years.49
Although it has traditionally been held that serology tends to be negative in malignant syphilis, in 1 study of 35 cases of this variant of secondary syphilis dating back to 1930, positive antibodies were identified in all cases. Furthermore, no correlation was observed between antibody titers and either disease severity or duration during follow-up.50
The following factors may help to identify malignant syphilis: strongly positive rapid plasma reagent titers, severe Jarisch-Herxheimer reactions, clinical and histological findings, and rapid resolution of lesions with appropriate antibiotic therapy. Malignant syphilis shares histologic features with other forms of secondary syphilis. Common findings include epidermal necrosis, a dense lymphoplasmacytic inflammatory infiltrate in the papillary and mid dermis, and a superficial perivascular infiltrate. There have also been reports of multinucleated giant cell reactions, and vascular endothelial changes in the dermis in the form of inflammation, proliferation, and fibrinoid necrosis.50
Immunohistochemistry studies and PCR can detect T pallidum in the tissue of patients with negative serology or microscopic examination, with some studies describing a sensitivity of around 92% for the combined use of these 2 techniques.51
Tertiary SyphilisTertiary syphilis is characterized by the presence of few treponemes but strong reactivity to these. A high index of suspicion is required as this diagnosis is extremely rare. Classical manifestations may change following the administration of antibiotics for another reason.
The most common clinical forms in tertiary syphilis are neurosyphilis (in particular general progressive paralysis and tabes dorsalis), cardiovascular syphilis (in particular syphilitic aortitis), and gummatous syphilis.
The size of the gummas is highly variable, but severe lesions, also known as rupioid lesions, have been described in malnourished patients.
When gummas grow on internal organs, they manifest as granulomatous masses, which can require the exclusion of other diseases, in particular sarcoidosis.
In one recently described case, a patient developed cavernous sinus syndrome as an atypical presentation of gummatous syphilis.52
Although neurosyphilis was once believed to be a late manifestation of T pallidum infection, it is now accepted that it can occur at any stage of the disease. A recent report described the case of an HIV-seronegative patient with an atypical combination of general progressive paralysis, meningovascular syphilis, and syphilitic meningitis; the patient also developed psychotic symptoms followed later by status epilepticus and limb weakness.53
Characteristics of Syphilis and HIV CoinfectionSince the outbreak of the HIV epidemic in the 1980s, there have been numerous isolated reports of syphilis and HIV coinfection with atypical, aggressive manifestations of syphilis particular to each of the different stages of the disease.48,54 More recent studies of larger series of patients have reported certain clinical differences between syphilis patients with or without HIV coinfection, but the overall conclusion is that that the general manifestations are similar at each stage, regardless or not of the presence of HIV infection.55
While chancres are generally solitary in primary syphilis, the possibility of multiple chancres is greater in patients with HIV coinfection (Fig. 1).54,56
Secondary syphilis manifestations generally appear within 3 to 6 weeks of resolution of the primary chancre, but there have been many reports of the overlapping of primary and secondary syphilis manifestations in patients with HIV coinfection.54 Accordingly, it is not rare to see cases in which patients with a primary chancre have simultaneous manifestations of secondary syphilis.
Patients with HIV coinfection typically have severe, persistent cutaneous manifestations,48 such as malignant syphilis and other atypical forms. The fact that most patients with malignant syphilis and HIV infection do not have severe immunosuppression (CD4 counts are typically >200 cells/mL)57–59 suggests that the immune dysfunction is more qualitative than quantitative.60 In 1 patient with malignant syphilis and HIV infection, histologic examination revealed a noncaseating granulomatous infiltrate with plasmacytic and histiocytic cells in which spirochetes were isolated.61
T pallidum invades the CNS both frequently and early on in patients with neurosyphilis and HIV coinfection.62 In 1 series, 13% of 455 patients with syphilis and HIV coinfection had ocular syphilis (a form of neurosyphilis) as the presenting clinical manifestation.63,64
In patients with tertiary syphilis and HIV coinfection, atypical presentations have included gummas in the CNS65,66 and gummas in early stages of infection.65 There have also been notable reports of rapid progression of syphilitic aortitis in HIV-positive patients; this finding is very uncommon in the absence of HIV infection.67
As mentioned previously, early CNS infection by T pallidum appears to be common in patients with HIV infection. One study proposed the following risk factors for neurosyphilis in patients with HIV: a CD4 count of over 350 cells/μL, an RPR titer of 1:128, and male sex.68
In another study of 117 patients with HIV and neurosyphilis, 33% of patients had asymptomatic neurosyphilis.62 There have also been reports of otosyphilis in HIV patients with CNS abnormalites.69
Several cases of ocular syphilis resulting in blindness have been recently reported in MSM. This condition is now considered to be the most common manifestation of neurosyphilis in HIV-infected individuals and appears to be associated with immune reconstitution inflammatory syndrome.63,64,70,71
Syphilis is diagnosed in the same way in HIV-positive and HIV-negative patients. Although experts largely agree that interpretation of serological tests is the same in both cases, there have been reports of serological anomalies in HIV-positive patients. These include unusually high titers, false-negative results, and abnormally delayed seroreactivity.72
In 1 study of 341 patients with syphilis, RPR titers were significantly higher in patients with HIV coinfection than in those without, but no differences were observed in clinical stages between the 2 groups.73 A higher frequency of biological false-positive results has also been described in patients with HIV infection.72,74
Nevertheless, false-negative results are more common in HIV patients with severe immunosuppression.75 Alternative diagnostic tests, such as skin biopsy, should be performed in HIV-infected individuals with symptoms suggestive of syphilis but negative serology.
ConclusionsSyphilis has multiple atypical cutaneous manifestations with which dermatologists need to be familiar. Deviations from classical clinical patterns not only pose diagnostic challenges but can also result in delayed treatment.
Severe, persistent, and atypical cutaneous manifestations of syphilis tend to be more common in patients with HIV coinfection.
Considering the high rate of HIV coinfection—and hence immunosuppression—in patients with syphilis, it is important to note that atypical clinical manifestations can facilitate the spread of both infections.
The steady rise in syphilis cases over recent years, particularly in individuals who engage in at-risk behaviors, requires vigilance on the part of practitioners and a high index of suspicion when faced with atypical clinical manifestations and/or manifestations that have not responded to treatment.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
We would like to thank Dr Agustín España for his contribution to this review.
Please cite this article as: Lleó MI, Escribano PC, Prieto BM. Manifestaciones cutáneas atipícas en la sífilis. Actas Dermosifiliogr. 2016;107:275–283.