Malignant lentigo, or lentigo maligna (LM), is a variant of melanoma in situ that develops mainly on chronically exposed areas of the skin in elderly patients.1 It accounts for 79% to 83% of all cases of melanoma in situ; in contrast with other variants of this condition, its clinical course is prolonged—sometimes several decades—before progressing to the invasive form, lentigo maligna melanoma (LMM).1–3
In Spain, the group of melanomas that includes LM and its invasive variant accounts for 15% of cases, probably as a result of the agriculture-based society in which most current patients lived.4 It affects both sexes equally, and approximately 50% of cases are LM. Incidence is estimated to be approximately 0.5 cases per 100 000 inhabitants per year. LM/LMM is diagnosed at a median age of 70 years, and 86% of cases are located on the head or neck.4
Clinically, LM is a macule that usually has several asymmetrically distributed brown tones. Its margins are irregular and poorly defined, and it grows slowly. Histologically, LM is composed of a proliferation of atypical melanocytes distributed individually along the dermoepidermal junction in an atrophic epidermis. Malignant melanocytes characteristically extend along the follicular epithelium. The dermis usually contains marked solar elastosis, as well as a lymphocytic infiltrate of varying intensity.1
The treatment of choice for LM is surgery.2 It is currently recommended that melanomas are resected in situ with a safety margin of 0.5cm. However, this distance may be insufficient to avoid involvement of the margins, and further surgery is necessary.3 Consequently, all surgical margins should be mapped as part of a thorough assessment. For many authors, modified Mohs surgery (with deferred assessment of margins in paraffin-embedded samples) is the approach of choice.3,5
LM usually presents in elderly patients, sometimes with concomitant conditions that can make surgery difficult to perform or even contraindicate it. As it generally presents on the face, aesthetic and functional aspects must be taken into account. Alternative approaches include nonsurgical options, such as radiotherapy, cryotherapy, or imiquimod.2,6,7
It is difficult to establish the real limits of LM. In addition, when deciding on the most appropriate therapy, a series of important factors must be taken into account. First, LM progresses to LMM in 5% to 50% of cases. These percentages vary so widely because published studies report results with very different lengths of follow-up and with the considerable selection bias associated with non–population-based hospital series.2,3 Second, although this lesion has the potential to progress to an invasive form, there are currently no clinical, histological, or biological data that enable us to predict whether it will do so. Third, and particularly relevant when selecting the optimal treatment, 22% of lesions previously diagnosed as LM after incisional biopsy yield an invasive component in the histopathology study.2
Imiquimod is a topically applied immunomodulator. By stimulating toll-like receptors 7 and 8, it can effectively enhance the innate and acquired immune responses and thus make it possible to treat skin tumors and lesions of viral origin.8 The innate response involves maturation and secretion of cytokines by antigen-presenting cells.8 Furthermore, activation of nuclear factor κB leads to increased production of cytokines by type 1 T helper cells, including tumor necrosis factor α, interleukin 12, interferon α, and interferon γ. Consequently, CD8 cells are activated and become cytotoxic T cells that destroy tumor cells. In addition, imiquimod induces apoptosis in tumor cells.8 The Spanish health authorities have approved it for the treatment of superficial basal cell carcinoma, actinic keratosis, and external genital warts.
Imiquimod is a particularly attractive option for the treatment of various tumoral lesions, thanks to its mechanism of action, efficacy, and excellent aesthetic outcome; moreover, it has been widely used in the approved indications and obviates the need for surgery. Consequently, since it was first launched, imiquimod has been used off-label for many other types of tumor, with varying clinical responses.Use of this agent to treat LM was first reported in the year 2000 as an alternative to surgery for a lesion in an elderly patient in poor general health who refused to undergo surgery.9 The drug was administered for 7 months at different frequencies that were modified according to the degree of local inflammation (from 1 daily application 3 times weekly to 2 daily applications). A biopsy specimen was obtained at the end of treatment, and the patient experienced no clinical relapses during the 9 months of follow-up. Since then, 234 patients have been treated with imiquimod in uncontrolled studies; 161 of these cases were recently reviewed by Erikson et al.2,10–16
Careful reading of these studies reveals a series of important points. First, therapy protocols vary widely, as does assessment of response; in addition, the studies range from case reports or very small series to open-label studies with up to 48 cases.2,10–16 Dosage varies from 3 times weekly to daily, with a duration of 2 weeks to 7 months, and follow-up is short, with a median of less than 24 months.2
Second, the evaluation of the response of LM to imiquimod must be taken into account. Erikson et al2 showed that the biopsy-confirmed clinical response rate was 88%. However, this rate is probably lower, given the manner in which the histological confirmation of clinical cure was evaluated. In most cases, the specimen was obtained by 1 or 2 punch biopsies in areas with clinical evidence of a lesion. Therefore, as not all the treatment area was evaluated, the positive predictive value and negative predictive value of the clinical evaluation of response could not be properly calculated. The only study to evaluate the histological response by means of total resection of the lesion revealed complete cure in no more than 75% of cases (30 cases in a series of 40 patients) with a negative predictive value of 91%; 30 of the 33 clinically cured cases were confirmed by biopsy.17 Important, even in this work, not all the specimen was studied: after resection of the area with the initial lesion using Mohs surgery, a conventional analysis was made of the central area, which represents only 5% of the specimen; therefore, both the response rate and negative predictive value could be even lower. Furthermore, an invasive component was present in 1 of the 3 cases with no clinical evidence of a lesion and biopsy-confirmed LM.17
Finally, the literature contains 3 cases in which progression of LM to LMM is described during treatment with imiquimod or once it was clear that a clinical response was not forthcoming.12,15 In 1 of the cases, the tumor reached a thickness of 3.3mm and developed satellitosis. In theory, imiquimod could have induced the invasive capacity of the melanoma, as it increases production of tumor necrosis factor α, which in turn stimulates production of metalloproteinase 9, a factor contributing to the invasive capacity of melanoma. In any case, treatment of LM with a clinically occult invasive component entails a proven risk that could favor tumor progression in patients receiving imiquimod.2
Therapy with imiquimod can also be analyzed from another angle. Today, 10 years after the first case of treatment of LM with imiquimod was reported, it is surprising that this is still an off-label indication and that there has been a continuous flow of publications comprising isolated cases or short series without providing any scientific evidence of its real efficacy and safety profile, as discussed above. In contrast, some dermatologists are still somewhat reluctant to use imiquimod in indications approved by the Spanish Agency for Medicines and Health Products. It is worth remembering that, for a drug to obtain approval for a given indication, it must have undergone a long and rigorous evaluation process involving controlled clinical trials that demonstrate beyond doubt the drug's efficacy and safety profile, as was the case for treatment of superficial basal cell carcinoma and actinic keratosis. Therefore, administration of imiquimod to treat LM should be more carefully evaluated before this agent can be added to the therapeutic arsenal. Although some studies have used dermoscopy to follow patients treated with imiquimod and as a complement to clinical evaluation of response, scientific evidence of its value has not been suitably confirmed.18–20 Confocal microscopy is more promising: this technique could enable us to perform studies that are more reliable in terms of assessment of response and inclusion of cases in which invasion has been completely ruled out.21
Finally, radiotherapy remains a valid alternative for the treatment of LM that cannot be treated with surgery, and cure rates ranging from 86% to 95% have been reported.2,6,22 The largest study included 96 patients with LM and 54 with LMM.22 Response was complete in all cases and recurrence was observed in 5/96 cases of LM and 2/54 cases of LMM after follow-up of at least 2 years. In another study of 64 patients with LM and LMM (in whom the invasive nodule had been resected), no relapses were observed after 2 years of follow-up.22 Furthermore, in terms of patient comfort, the recommended radiotherapy regimen involves administration of 100Gy over 10 sessions (5 weekly sessions for 2 weeks); therefore, despite the presence of acute radiodermatitis, the lesions remain for a much shorter period than those reported in any of the regimens evaluated for imiquimod.6 Furthermore, radiotherapy has proven efficacious for the treatment of the invasive component; therefore, it would be suitable for the 22% of cases of LM that actually involve invasion.2,22
Cryotherapy has also been used as an alternative in cases where surgery is contraindicated. Although relapse rates (6.6%-8.3%) are similar to those of radiotherapy, the possibility of unwittingly treating invasive melanoma, together with reports of cases progressing to invasive melanoma after treating LM with cryotherapy, advises against its use, except in special situations where no alternatives are available.23,24
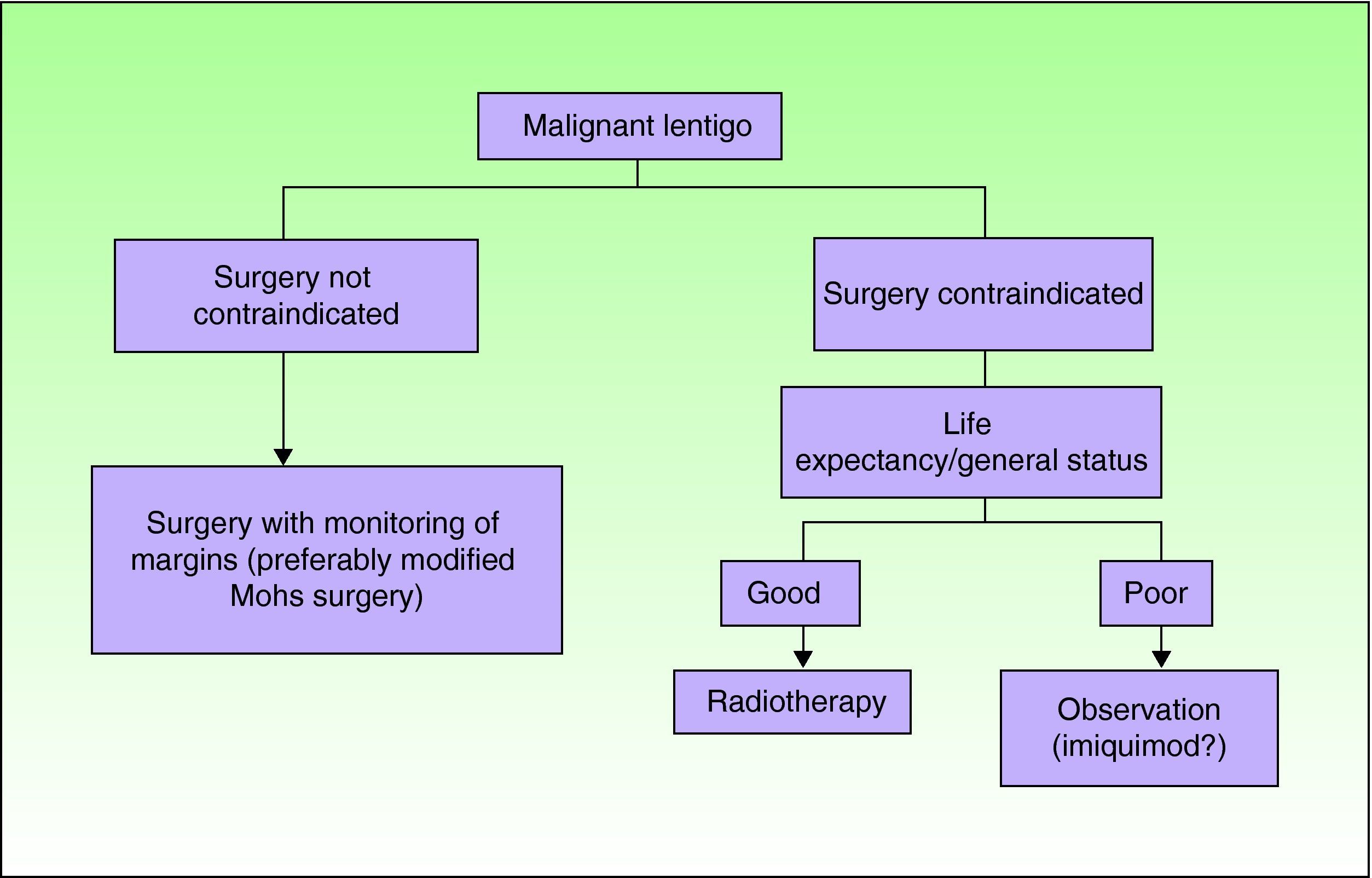
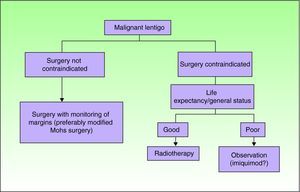
Figure 1 shows the proposed algorithm for the treatment of LM. Although not excluded as an option, imiquimod is reserved for very specific and selected situations in which patients or their families have been given appropriate information about the scientific evidence for its use. In fact, British guidelines recommend applying it only in clinical trials.25 Where feasible, the option should be discussed by a tumor committee. The possibility of adopting a conservative approach with suitable clinical follow-up should not be ruled out in some of these cases. Indeed, some expert groups consider this approach a valid option.
To conclude, surgery is the treatment of choice in LM. In particular, margins should be carefully monitored, as in modified Mohs surgery. At present, imiquimod should not be considered first-line treatment in patients who cannot undergo surgery. Use of this agent should be restricted to those cases in which alternative treatments (eg, radiotherapy) cannot be administered, at least until the clinical methods applied to evaluate it are improved, for example, through use of confocal microscopy. We can then be sure we are treating true LM, with no occult invasion. More robust scientific evidence of its efficacy and safety profile is also essential.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as. Nagore E, et al. Imiquimod para el tratamiento del lentigo maligno. Actas Dermosifiliogr. 2011;102:559-62.