Leishmaniasis encompasses a group of diseases caused by protozoa of the genus Leishmania. These infections affect millions and are endemic in the Mediterranean, North Africa, the Middle East, India, and Central and South America. Three clinical variants are distinguished on the basis of the culprit species and the patient's immunity: a cutaneous form is confined to the skin, a mucutaneous form affects both skin and mucosal tissues, and a reticuloendothelial form affects organs. Some three quarters of new cases are cutaneous, and although they may resolve spontaneously, choice of treatment is important because disfiguring cribriform scarring is common.1
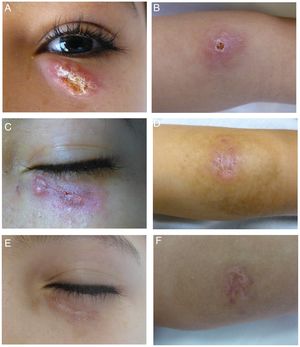
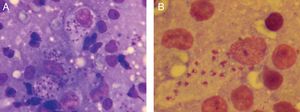
A 10-year-old boy originally from Morocco who had been living in Spain for 2 months was brought for examination of an asymptomatic 3-month-old lesion on a lower eyelid. He was in good health and had no systemic symptoms. Physical examination revealed an oval erythematous plaque with slightly infiltrated borders and a firmly adhered crust in the center (Fig. 1A). A similar plaque was found on his left forearm (Fig. 1B). The locations of the lesions in exposed areas of skin in a patient from a geographic area where Leishmania species are endemic led to a diagnosis of cutaneous leishmaniasis (CL). Microscopic examination of a Riu-stained skin smear revealed macrophages with amastigotes, confirming the diagnosis (Fig. 2). Polymerase chain reaction (Spanish National Microbiology Center, Instituto de Salud Carlos III) was used to identify the species as Leishmania major.
A–B, Lesions caused by Leishmania major before treatment. Note the erythematous oval plaque, the slightly infiltrated borders, and the central crusting on the lower eyelid and left forearm. C–D, Clinical response to 4 sessions of photodynamic therapy. E–F, Response 3 mo after treatment ended.
We evaluated available treatments and started photodynamic therapy (PDT) with methyl amino-levulinate as the photosensitizing agent. Each session started with removal of the crust and application of the photosensitizer on the lesion and 5mm around it. After a period of occlusion (3 hours), each site was irradiated with narrow spectrum (70–100 mW/cm) visible red light (Aktilite CL128, Galderma, Lausanne, Switzerland) at a peak of 630nm and a total energy level of 37J/cm2. The eyes were protected with shells (Spectraview Shield, Sperian, Honeywell, Morris Plains, NJ, USA), and the light was adjusted to fall near the free border around the eyelid, leaving a nonirradiated distance of 3mm at the upper border. PDT was repeated weekly until clearing was complete after 7 sessions and only residual signs could be observed. Three months after treatment ended there was a superficial hypopigmented scar (Fig. 1, C–F). The main adverse effect was slight to moderate pain. The patient tolerated the pain well when we interrupted irradiation for brief intervals, during which the area was air-cooled.
Choosing a treatment for CL is challenging because of the scarcity of evidence analyzing risks and benefits. Various guidelines have been published but there is no international consensus. The Infectious Diseases Society of America (IDSA) has established clinical criteria for identifying cases with the worst prognosis (Table 1). Once a risk group has been determined, the physician can consider systemic treatments for complicated variants and topical alternatives for uncomplicated ones.2 If we had followed this approach strictly, the facial lesion would have been eligible for systemic treatment with miltefosine, azoles, pentavalent antimonials, amphotericin B, or pentamidine. All these drugs have potential adverse effects of various types.3 After consideration of our patient's young age, the presence of only 2 isolated lesions, and the facial location of one of them as the only criterion for categorizing the case as complicated, we ruled out systemic treatment. The nonsystemic alternatives available included intralesional antimonial injection, which is relatively contraindicated around the eyelids and in acral zones; paromomycin; and PDT.
Clinical Features That Affect the Treatment of CL2.
| Uncomplicated CL | Complicated CL |
|---|---|
| Infection by species not associated with mucosal involvement | Infection by species associated with mucosal involvement |
| No mucosal involvement | Subcutaneous nodules |
| No signs of complicated CL | Large, swollen lymph nodes |
| Single or few lesions | More than 4 lesions >1cm in diameter |
| Small lesion (diameter, <1cm) | Single lesion >5 cm |
| Location accessible for topical treatment | Location or size inappropriate for topical treatment |
| Aesthetically unimportant location | Lesions on the face (including ears, eyelids, and lips), fingers, large toe, or genitals |
| Immunocompetent patient | Immunocompromised patient |
| Lesion that resolved without treatment | Failure of topical treatment at 2–3 mo |
Abbreviation: CL, cutaneous leishmaniasis.
According to the summary of product characteristics for the PDT material, this modality is indicated for treating actinic keratosis, Bowen disease, and certain basal cell carcinomas.4 Approval has also been given in recent years for treating skin infections that are refractory to conventional treatment.5,6 The germicidal effect of porphyrins was described in 1988 when Leishmania parasites disappeared from macrophages on exposure to hematoporphyrin and menadione.7 Based on these findings the first use of PDT with aminolevulinic acid for CL was described in 2003.8 One patient with multiple lesions, half of which provided the controls, received topical paromomycin on 5 lesions and PDT on the other 5. Better and faster results were achieved with PDT. In the first randomized clinical trial comparing PDT to paromomycin, published in 2008, all patients treated with PDT improved and over 90% experienced complete clearing.9 Although our patient's tolerance to pain on irradiation was at an acceptable level, greater tolerance can be achieved with daylight PDT, which has been proposed as an alternative when conventional facilities for PDT are unavailable.10 Daylight PDT can be self-administered and is less painful.
In summary, PDT is a safe, less aggressive treatment that does not generate resistance and can achieve full clearance of CL lesions. It leaves residual scars that are not depressed and provides optimal long-term aesthetic results. These characteristics make it an effective alternative treatment for CL in our clinical practice.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Sainz-Gaspar L, Rosón E, Llovo J, Vázquez-Veiga H. Terapia fotodinámica en el tratamiento de la leishmaniasis cutánea. Actas Dermosifiliogr. 2019;110:249–251.