Hansen disease, or leprosy, is caused by Mycobacterium leprae or Mycobacterium lepromatosis. Because these bacteria enter the body via the upper airways, they generate clinical manifestations in the nasal mucous membranes and the mouth. We aimed to describe the characteristics of oral lesions in patients with Hansen disease.
Material and methodsCross-sectional observational study of 100 patients diagnosed with Hansen disease. We examined the oral cavity and recorded clinical findings on a disease reporting form for each patient. We also included the histopathologic findings for lesions that required a biopsy. Samples suggestive of Hansen disease were processed with hematoxylin-eosin, Ziehl–Neelsen, and Fite–Faraco staining. Variables were analyzed, as appropriate, with the χ2 test, the Mann-Whitney U test, or the Spearman correlation coefficient.
ResultsSixty-seven men and 33 women were included. The mean (SD) age was 48.1 (16.4) years. Thirty-four patients had oral lesions. Lesions related to Hansen disease were found in 9 patients. The locations were the hard palate and upper lip. Oral lesions were significantly more frequent in patients with lepromatous leprosy, borderline lepromatous leprosy (P = .029), and erythema nodosum leprosum (P = .031).
ConclusionsThe frequency of oral lesions is low in Hansen disease. Such lesions present as lepromas and leprous plaques on the hard palate and upper lip.
La enfermedad de Hansen o lepra es una enfermedad causada por Mycobacterium leprae y Mycobacterium lepromatosis. Estas bacterias ingresan al organismo por las vías aéreas superiores, por lo que generan manifestaciones clínicas en la mucosa nasal y en la cavidad bucal. El objetivo del estudio es caracterizar las lesiones bucales de los pacientes con una enfermedad de Hansen.
Materiales y métodosSe diseñó un estudio observacional, transversal, de 100 pacientes con diagnóstico de lepra. Se examinó la cavidad bucal y se registraron los hallazgos clínicos en una ficha epidemiológica. Además, se incluyó el estudio histopatológico de las lesiones que requirieron de una biopsia. Los casos sugestivos de lepra fueron teñidos y analizados con hematoxilina y eosina, Ziehl-Neelsen y Fite-Faraco. Las variables fueron analizadas y Fite-Faraco. Las variables fueron analizadas y comparadas mediante las pruebas Chi2, correlación de Spearman y U de Mann-Whitney.
ResultadosEl grupo de estudio estuvo conformado por 67 hombres y 33 mujeres, con una media (± desviación estándar) de 48,1±16,4 años de edad. Fueron observadas lesiones bucales en 34 pacientes. Las lesiones orales relacionadas con lepra, presentes en 9 pacientes, se ubicaron en el paladar duro y el labio superior. Estas fueron significativamente más frecuentes en la lepra lepromatosa, la borderline lepromatosa (p=0,029) y en el eritema nudoso leproso (p=0,031).
ConclusionesLas lesiones bucales asociadas a lepra ocurren con una frecuencia baja, manifestándose como lepromas y placas leprosas en el paladar duro y el labio superior.
Leprosy is considered a public health problem, especially in certain developing countries. Its prevalence has decreased notably since the introduction of multidrug therapy, but high numbers of cases are still recorded in countries in Southeast Asia, Latin America, Africa, the eastern Pacific, and the western Mediterranean. India is the country with the highest prevalence in the world, followed by Brazil.1–4
Leprosy is a chronic infectious disease that mainly affects the skin and peripheral nerves, and is caused by the acid-fast bacteria Mycobacterium leprae and Mycobacterium lepromatosis. However, there can be considerable clinical variability among patients with leprosy, which can also affect the eyes, mucous membranes, and bones.1–4
Depending on clinical manifestations, the World Health Organization classifies leprosy into 2 broad categories: paucibacillary leprosy (1–5 lesions on the skin and/or 1 affected nerve trunk) and multibacillary leprosy (6 or more lesions on the skin and 2 or more affected nerve trunks).1,4
The Ridley-Jopling classification1 categorizes leprosy as tuberculoid (TT), borderline tuberculoid (BT), borderline-borderline (BB), borderline lepromatous (BL), lepromatous (LL), and indeterminate (IL) leprosy. If the patient lacks a cellular immune response, lepromatous leprosy develops. Patients with an effective cell-mediated immune response tend to develop the tuberculoid form. The borderline forms fall between the 2 aforementioned forms, occurring in patients with varying degrees of immune response. Lastly, indeterminate leprosy is considered the initial stage of the disease.3,5
Leprosy reactions are immunological events that can occur before, during, or after treatment of the disease. Type 1 leprosy reactions are in turn divided into degradation reactions if there is a transition towards the lepromatous pole, and reversal reactions if there is a transition towards the tuberculoid pole. Type 2 leprosy reactions are subdivided into erythema nodosum leprosum, erythema polymorphic, and Lucio phenomenon or diffuse leprosy.
The clinical variability of leprosy is determined by the tropism of the microorganism for the skin and peripheral nervous tissue, the cellular immune response, and the patient’s susceptibility to mycobacteria. Depending on the type of leprosy, cutaneous manifestations vary in terms of both lesion number and distribution.7 The majority of patients develop LL, which is a multibacillary form, characterized by multiple nodules on the skin and mucosa (lepromas). The most common locations of these lesions are the face and pinna.7 BL and BT leprosy are identified in a smaller proportion of patients. These patients are in an unstable stage, which eventually evolves to one pole or another on the leprosy spectrum. Clinically, BT leprosy presents as annular erythematous plaques with diffuse external borders and well-defined internal borders. The BL form is characterized by the presence of multiple hypopigmented papules, nodules, and infiltrated plaques.7,8 The higher frequency with which the BL and LL forms are identified can be attributed to an imbalance of the immune system at the moment of the clinical examination and the higher probability of polarizing to the LL form.9,10
Up to 30% of patients with a diagnosis of leprosy develop severe acute exacerbations either spontaneously or during treatment.9 These exacerbations are referred to as leprosy reactions and reflect a sudden deterioration of the immunological balance between the pathogen and the infected individual. Leprosy reactions can occur even after successful treatment.1,7 Erythema nodosum leprosum was the most common leprosy reaction in our study, followed by reversal phenomena in multibacillary forms of the disease.
Reversal reactions correspond to an increase in cellular immunity against M leprae. They are characterized by inflammation, pain in existing skin lesions, and neuritis. Type 2 leprosy reactions correspond to a systemic inflammatory response to immune complexes and are characterized by tender skin nodules, fever, and inflammation of organs (liver, lungs, joints, and eyes).11 In accordance with our findings, it has been reported that leprous erythema nodosum is the most prevalent lesion in multibacillary leprosy patients and its presence, course, and persistence may be associated with odontogenic infections and periodontal disease.12
It has been suggested that the clinical examination of patients diagnosed with leprosy should include the oral mucosa, as this may constitute a second route of mycobacterial transmission and infection. Oral lesions are usually asymptomatic and secondary to nasal changes. They are infrequent and are usually associated with LL or reactional phenomena. Another route of infection in the oral mucosa involves the hematogenous or lymphatic spread of the bacillus, favored by the temperature of the upper airways.
The oral manifestations described to date are varied, and include macules, papules, nodules that can ulcerate or necrotize, and bone changes.12–14 Hypopigmentation of the mucosa, atrophy of the lingual papillae, and retraction of the uvula have also been described.1,13
In contrast to leprosy-related skin lesions, which are well documented in the literature, few studies have characterized lesions in the oral cavity directly related with this disease, possibly due to their low prevalence.12–14 Furthermore, nonspecific lesions that occur in some leprosy patients can also manifest in the general population.15,16 The objective of the present study was to characterize oral lesions present in patients with Hansen disease.
Material and MethodsThis cross-sectional observational study included 100 patients who were diagnosed with Hansen disease and treated at the Dr. Jacinto Convit Institute of Biomedicine in Caracas, Venezuela. An epidemiological file, validated by experts, was used to record patient personal data and medical history, as well as data on disease course and treatment.
A systematic examination of the oral cavity was conducted. Depending on the nature of the lesions and their relationship with the provisional diagnosis, a biopsy was taken for histopathological study. In the case of lesions associated with suspected Hansen disease, Fite-Faraco and Ziehl Neelsen staining were performed for identification of mycobacteria.
The study was approved by the Bioethics Committee of the School of Dentistry, Universidad Central de Venezuela (CB-059-2018). Patients received information on the nature of the examination and the procedures to be followed, and clarification of any doubts regarding the procedure. All patients provided written informed consent. The results were shared with the research participants and all lesions were treated. Confidentiality was guaranteed.
The descriptive analysis was carried out using percentages of absolute values and the mean ± standard deviation (SD). Correlations between variables were examined using the χ2 test, Spearman correlation analysis, and the non-parametric Mann-Whitney U test. A P-value <0.05 was considered statistically significant.
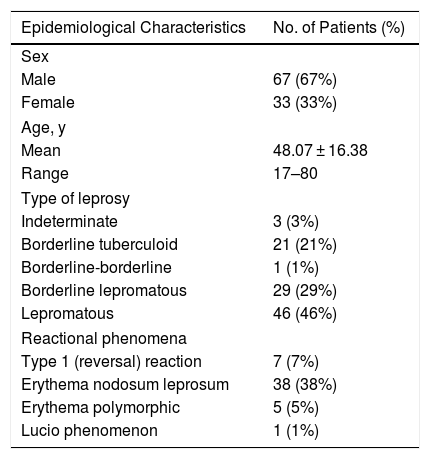
ResultsThe epidemiological characteristics of the evaluated sample (n = 100) are summarized in Table 1.
Epidemiological Characteristics of 100 Patients With Hansen Disease
| Epidemiological Characteristics | No. of Patients (%) |
|---|---|
| Sex | |
| Male | 67 (67%) |
| Female | 33 (33%) |
| Age, y | |
| Mean | 48.07 ± 16.38 |
| Range | 17–80 |
| Type of leprosy | |
| Indeterminate | 3 (3%) |
| Borderline tuberculoid | 21 (21%) |
| Borderline-borderline | 1 (1%) |
| Borderline lepromatous | 29 (29%) |
| Lepromatous | 46 (46%) |
| Reactional phenomena | |
| Type 1 (reversal) reaction | 7 (7%) |
| Erythema nodosum leprosum | 38 (38%) |
| Erythema polymorphic | 5 (5%) |
| Lucio phenomenon | 1 (1%) |
Ages ranged from 17 to 80 years (mean ± SD, 48.1 ± 16.4 y). Men predominated, and the most frequent type of leprosy was LL, followed by BL.
Reactional phenomena were observed in 51 patients: leprous erythema nodosum was the most frequent (30 diagnosed LL patients and 8 BL patients).
Presence of oral lesions in patients with Hansen diseaseOral lesions were observed in 34 patients. These lesions were not associated with leprosy in 25 patients and were associated with leprosy in 9. Of these 34 patients, 18 (55.9%) were men and 16 (44.1%) were women.
When the groups were stratified by lesion type, men outnumbered women in the group with oral leprosy-related lesions. Lesions unrelated to leprosy were observed in both men and women (Table 2). Although more men had oral lesions (regardless of their association with leprosy), this difference was not statistically significant (p = 0.454).
Of the 34 patients, oral lesions, whether associated or not with leprosy, were located on the hard palate in 12 (35.3%) and the upper lip in 8 (23.5%) (p = 0.001) (Table 3).
Relationship Between Type of Leprosy and Presence of Oral Lesions
| Leprosy Type | Patients Without Lesions, N | Patients With Lesions Unrelated to Leprosy, N | Patients With Leprosy-Related Lesions, N | Total, N |
|---|---|---|---|---|
| LI | 2 | 1 | 0 | 3 |
| BT | 14 | 7 | 0 | 21 |
| BB | 0 | 1 | 0 | 1 |
| BL | 17 | 9 | 3 | 29 |
| LL | 33 | 7 | 6 | 46 |
| Total | 66 | 25 | 9 | 100 |
BB, borderline-borderline; BL, borderline lepromatous; BT, borderline tuberculoid; IL, indeterminate leprosy; LL, lepromatous leprosy.
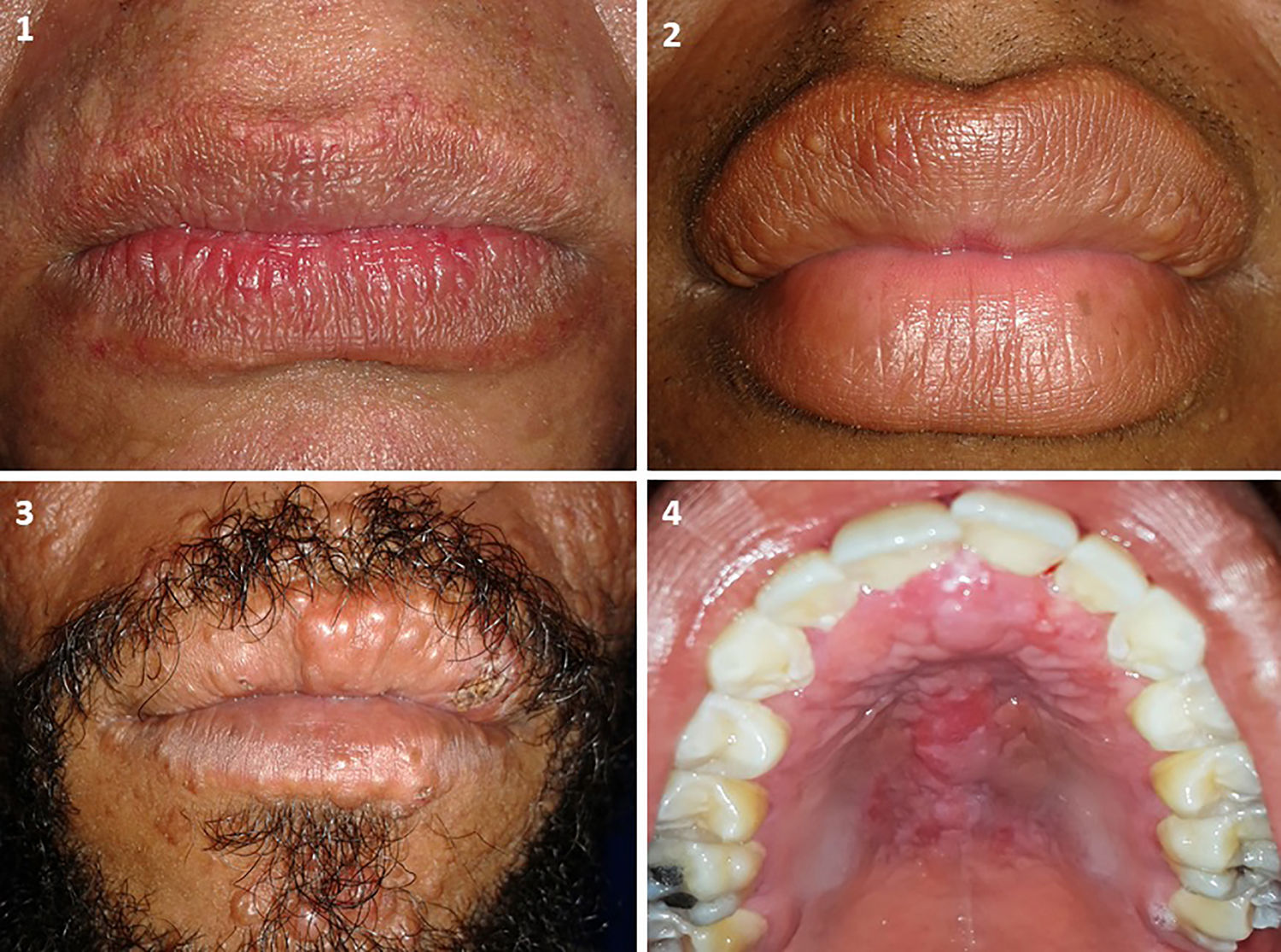
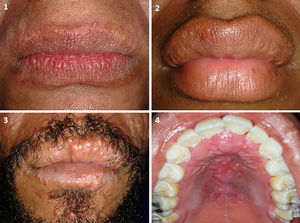
Leprosy-related lesions corresponded to leprous plaques in 5 of 34 patients (14.7%) and lepromas on the palate and upper and lower lips in 4 of 34 (11.8%) patients (Fig. 1). All leprosy-related lesions exhibited dense histiolymphocytic infiltrate, without the formation of granulomas. The bacterium was identified in all samples.
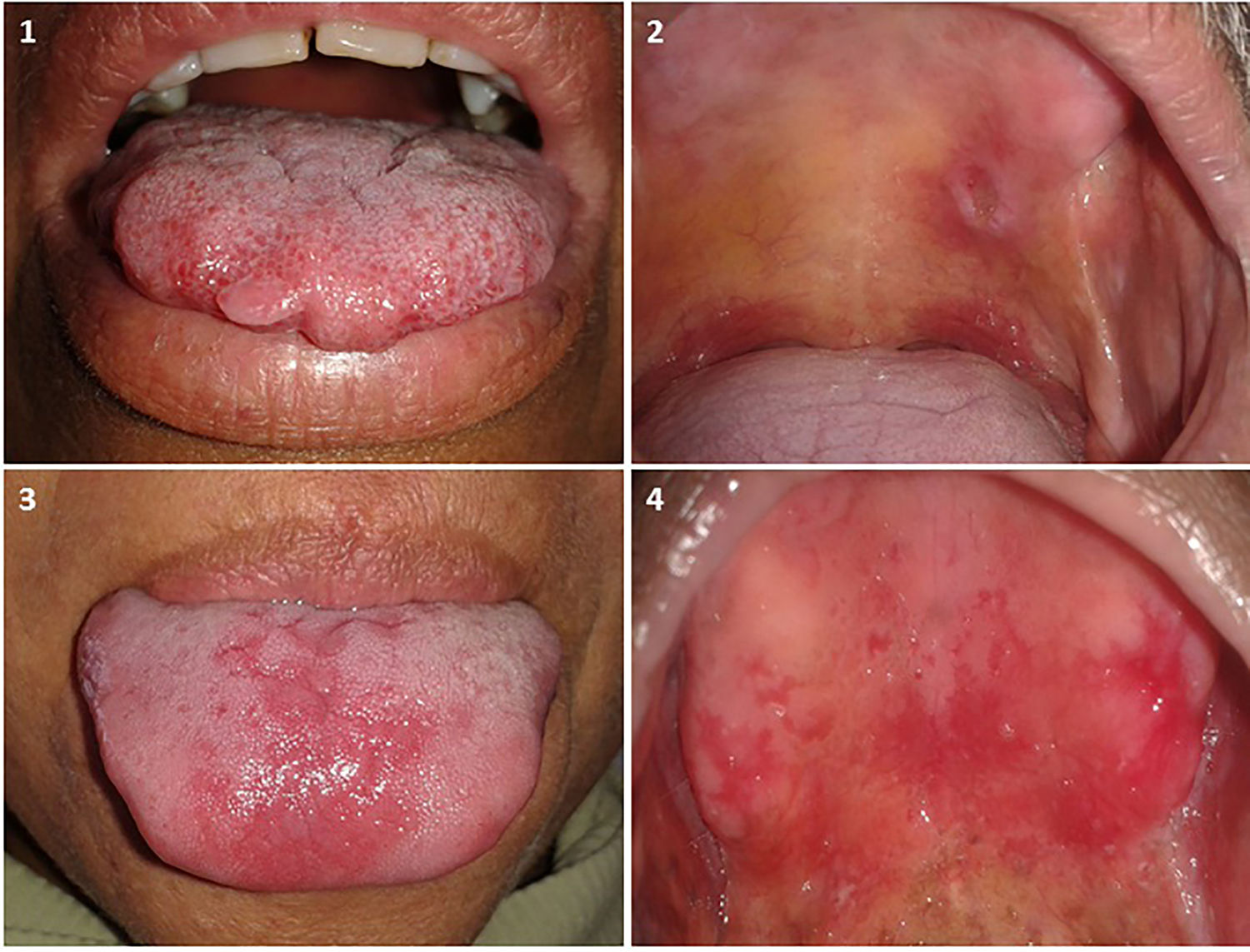
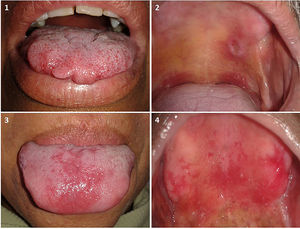
Of the 34 patients with lesions unrelated to leprosy, 7 (20.6%) had prosthetic stomatitis (PS), 6 (17.6%) had traumatic fibromas, 3 (8.8%) had melanotic macules, 2 (5.9%) had traumatic ulcers, and 3 (5.9%) had vascular malformations. In addition, lichen planus, melanocytic nevus, erythematous candidiasis, mixed lesions (leukoplakia, PS, traumatic ulcer), and mixed lesions including leprosy-related lesions (mucocele, leprous plaque) were diagnosed in isolated patients (2.9%) (Fig. 2).
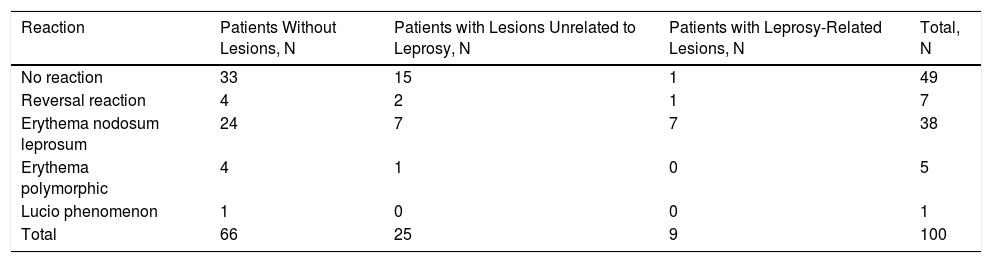
Of the 34 patients, 19 (55.9%) presented reactional phenomena, the most common of which in patients with oral lesions was leprous erythema nodosum (p = 0.031). Leprosy-unrelated oral lesions were observed in a higher percentage of patients without reactional phenomena (Table 4).
Reaction to Leprosy and Presence of Oral Lesions
| Reaction | Patients Without Lesions, N | Patients with Lesions Unrelated to Leprosy, N | Patients with Leprosy-Related Lesions, N | Total, N |
|---|---|---|---|---|
| No reaction | 33 | 15 | 1 | 49 |
| Reversal reaction | 4 | 2 | 1 | 7 |
| Erythema nodosum leprosum | 24 | 7 | 7 | 38 |
| Erythema polymorphic | 4 | 1 | 0 | 5 |
| Lucio phenomenon | 1 | 0 | 0 | 1 |
| Total | 66 | 25 | 9 | 100 |
At the moment of evaluation, 23 (67.6%) patients had completed multibacillary treatment, 10 (29.4%) were undergoing treatment, and 1 (2.9%) had yet to begin treatment. Although most of the lesions were observed during the treatment phase, there was no significant relationship between the presence of oral lesions and treatment phase (p = 0.057).
DiscussionThe frequency of oral mucosa involvement in patients diagnosed with leprosy is a topic of much debate in the literature, and ranges from 0% to approximately 60% of cases depending on the study consulted.17–19 In our study, lesions in the oral cavity were observed in 34% of patients evaluated.
In our study population, leprosy-related lesions were located on the hard palate and upper lip. Higher bacterial loads have been reported at lower temperatures.12 Moreover, according to the World Health Organization, the most affected locations in the oral cavity are the hard palate, the soft palate, and the maxillary gingiva.20
In patients with leprosy-related lesions, leprous plaques were observed at the boundary between the skin and the semi-mucosa of the lips. Lepromas (sessile papules of the same color as the adjacent skin located on skin and the semi-mucosa of the upper and lower lips) were also observed. On the palate, these appeared as nodules of the same color as the adjacent mucosa. Similar characteristics have been described in India in a patient with LL.21 In contrast to the results presented here, other authors have described diffuse hyperpigmentation, hypopigmentation, ulcers on the gums and cheeks, retraction of the uvula, and microstomia.15,18
Oral cavity lesions occurred with a higher frequency in patients with LL, consistent with previous reports associating these lesions with more advanced stages of the disease.6,13 The absence of lesions in the buccal mucosa of patients with TT and BT leprosy can be attributed to the fact that these paucibacillary forms of the disease are characterized by few lesions, which are usually limited to the skin, with no detectable bacilli. However, the BB and BL forms can change over time and the likelihood of lesion appearance increases with increasing proximity to the lepromatous pole.12,13,22
In contrast to the above, M leprae has been identified by immunohistochemistry and polymerase chain reaction in the mucosa of patients with both multibacillary and paucibacillary disease, suggesting that this mycobacterium is present in the oral mucosa with a high frequency, and implying that this area is a potential site of bacterial transmission and infection.22
Lesions not directly related to leprosy included reactive, infectious, and immunological lesions. Other authors have described atrophic tongue and coated tongue in leprosy patients, and biopsies have shown the presence of nonspecific chronic inflammation, epithelial hyperplasia, and lichen planus.15,17 The presence of reactive lesions could be related to a loss of sensitivity of the oral mucosa, which is characteristic of LL, coupled with chronic trauma, such as that caused by defective prostheses, sharp dental edges, bruxism, etc. However, this hypothesis is controversial: other authors23 who have evaluated the sensitivity of different areas of the oral mucosa in leprosy patients have concluded that altered sensitivity in the oral cavity may not be as common as described for skin lesions elsewhere on the body.
It is important to note that lesions that affect the oral mucosa of individuals who are apparently healthy or have other diseases can also appear in the oral mucosa of leprosy patients. Biopsy of any lesion with a doubtful diagnosis is therefore essential. Nonspecific lesions, without granulomas or acid-resistant bacilli, are frequent and occur in different clinical forms of the disease.24
ConclusionsLeprosy-related lesions in the oral cavity are uncommon, but can manifest as lepromas or leprous plaques on the hard palate and lips. These occur more frequently in multibacillary forms of the disease, specifically LL and BL.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Moronta Castellano G, Villarroel-Dorrego M, Crespo Lessmann L. Caracterización de lesiones bucales de pacientes con enfermedad de Hansen. Actas Dermosifiliogr. 2020. https://doi.org/10.1016/j.ad.2020.05.007