Oral contraceptives combine estrogen and progesterone to suppress ovulation. Synthetic forms are usually used. In dermatology, oral contraceptives are prescribed for 2 main reasons: to prevent pregnancy when teratogenic drugs must be taken and to treat skin manifestations of hyperandrogenism. Most oral contraceptives improve both acne and hirsutism, but the androgenic effect of progestogens—particularly if the contraceptive contains first- or second-generation progestogens—can trigger or exacerbate acne. One of the most serious side effects of oral contraceptives, thrombosis, is mainly caused by the estrogen component and its dose. If we mainly consider a contraceptive's thrombotic profile when prescribing, the choice would be to have 30μg or less of ethinyl estradiol combined with levonorgestrel. On the other hand, if our main objective is to treat signs of androgenization, we would prefer contraceptives containing progestogens with antiandrogenic effects.
Los anticonceptivos orales son fármacos compuestos por un estrógeno y un progestágeno, habitualmente sintéticos, que inhiben la ovulación. En dermatología los anticonceptivos orales se emplean con 2 indicaciones principales: para evitar el embarazo cuando empleamos fármacos teratógenos y para tratar manifestaciones cutáneas de hiperandrogenismo. La mayoría de los anticonceptivos orales mejoran tanto el acné como el hirsutismo, sin embargo el empleo exclusivo de progestágenos como anticonceptivos, especialmente de primera o segunda generación puede agravar o desencadenar un acné, debido a su efecto androgénico.
La trombosis es uno de los efectos secundarios más graves de los anticonceptivos orales y depende sobre todo de los estrógenos empleados y su dosis. Si atendemos solo al perfil trombótico, el anticonceptivo oral de elección debería ser el que combinara 30 o menos μg de etinilestradiol con el levonorgestrel. Por el contrario, si lo que queremos es tratar signos de androgenización, los anticonceptivos orales preferidos serán aquellos con progestágenos de efecto antiandrogénico.
Dermatologists prescribe contraceptives in women with 2 main indications. When we want to prevent pregnancy when using potentially teratogenic drugs, and to treat cutaneous manifestations of hyperandrogenism, i.e., hirsutism, acne, and androgenetic alopecia.1
When the indication is merely contraception, a wide variety of contraceptive methods may be used, from ineffective natural methods to barrier methods, such as condoms, diaphragms, and spermicides, surgical methods, such as tubal ligation, vasectomy, and tubal micro-inserts, copper IUD placement, and, finally, hormonal contraceptives administered orally, transdermally, intramuscularly, or vaginally (Table 1). In practice, as the teratogenic therapies most commonly prescribed by dermatologists are oral retinoids, with treatment that tends to last several months, it is more common to prescribe reversible and simple contraceptive methods, such as oral contraceptives (OCs).
Hormonal Contraceptives.
| Estrogens+gestagens |
| 1. Oral: daily regimen |
| Monophasic: continuous administration of estrogen and progesterone 21 days |
| Biphasic: first phase with a lower progesterone dose than the second phase |
| Triphasic: imitates the natural cycle with 3 phases of rising doses of progesterone |
| 2. Transdermal, patches: weekly regimen |
| 750mg ethynylestradiol+6mg norelgestromin (norgestimate, 3rd gen.) |
| 20mg ethynylestradiol/150mg norgestimate/daily - 3 weeks and 1 week rest |
| 3. Vaginal, vaginal ring: monthly regimen |
| 15mg ethynylestradiol and 120mg etonogestrel |
| Gestagens |
| 1. Oral, minipill: 75mg desogestrel - daily |
| 2. Injectable, intramuscular: 150mg medroxyprogesterone acetate - 3 months |
| 3. Subdermal implants: levonorgestrel or etonogestrel; duration, 5 years |
| 4. IUD, levonorgestrel: duration, 4–5 years |
The indication for treating hyperandrogenism with OCs is justified because almost all combined OCs (estrogen+progestogen) can improve acne and hirsutism to a greater or lesser extent; however, contraceptives with progestogens alone (especially first-generation and second-generation progestogens, which have a certain androgenic effect) may not help and may even worsen or trigger acne.2 The effect of combined OCs against acne and hirsutism is due to the fact that combined OCs suppress production of androgens in the ovaries that are mediated by luteinizing hormone (progestogens suppress secretion of this hormone), the estrogens increase production of sex hormone binding globulin in the liver, and both of these events reduce levels of circulating androgens.3 The progestogens also compete with 5α-reductase and the androgen receptor. It is estimated that between 10% and 20% of women present clinical signs of androgenization, such as seborrhea, acne, hirsutism, or alopecia. Furthermore, a subgroup of these women also presents menstrual abnormalities, obesity, insulin resistance, type 2 diabetes, and raised serum lipids.4
The first combined OC was introduced in 1960 (Enovid®), with a combination of an estrogen (mestranol 0.15mg) and a progestogen (norethynodrel 9.85mg). The first case of thrombosis linked to the use of OCs5 was described the following year and this association has been shown by many subsequent studies. In the following decades, new combinations of estrogens and progestogens were sold, in some cases in search of a better cardiovascular risk profile, in others in an attempt to boost the antiandrogenic effect of the progestogens, without compromising their contraceptive effect. The combined use of different molecules and doses of estrogens and progestogens has given rise to an enormous number of possible combinations, with different advantages and disadvantages, which, when added to the nonspecific mechanism of action of the hormones, has made the study of OCs somewhat laborious and unattractive for dermatologists. In this review, we will attempt to clarify as far as possible the advantages and disadvantages of each type of OC in a way that is practical and useful for our daily practice.
Estrogens and Progestogens Used as Oral ContraceptivesOCs are drugs made up of an estrogen and a progestogen, both synthetic in most cases, which inhibit ovulation, justifying their high contraceptive effectiveness, which is reversible when the drug is withdrawn.
The estrogen component of OCs may be synthetic estrogens, such as ethynylestradiol (the most commonly used) or mestranol, natural estrogens such as estradiol, estrone, or estriol, or equine estrogens such as equilin, equilenin, hippulin, or equine estrone. The dose of the estrogens is important because it largely determines the risk of thrombosis. In practice, the estrogen in the vast majority of OCs is ethynylestradiol, a high potency synthetic estrogen with doses that vary depending on the preparation, although the tendency is a preference for low doses (from 15 to 50μg). In recent years, some combinations have appeared on the market, in which the estrogen is the natural estrogen (estradiol valerate and 17b estradiol), which is much less potent than ethynylestradiol but is tolerated much better. In general, synthetic estrogens have a higher risk of thrombosis than natural estrogens and the latter are therefore preferred in the indication of hormone replacement therapy, as the cardiovascular risk in women increases with age.
The progestogen component of OCs is the ovulation inhibitor and is therefore responsible for the contraceptive action. All progestogens have antigonadotrophic activity (hence their contraceptive action), as well as progestogenic and antiestrogenic action. The different progestogens, however, differ from each other in their ability to interact with androgen receptors (pro-androgenic and antiandrogenic), glucocorticoid receptors, and mineralocorticoid receptors. Different generations of progestogens have been developed based on these characteristics, resulting in 3 large families: progesterone derivatives (medroxyprogesterone acetate, cyproterone, chlormadinone, and nomegestrol), spironolactone derivatives (drospirenone), and first-generation testosterone derivatives (norethisterone, norethindrone acetate, lynestrenol), second-generation testosterone derivatives (norgestrel and levonorgestrel), third-generation testosterone derivatives (norgestimate, gestodene, desogestrel, etonogestrel) and fourth-generation testosterone derivatives (dienogest). The successive generations of testosterone-derived progestogens have reduced the androgenic effect and have acquired antiandrogenic potential, which is an advantage for dermatologic indications. Unfortunately, however, their thrombogenic profile has worsened and it has been shown that third-generation testosterone-derived progestogens are more thrombogenic than those of the second generation.6 Of the progestogens chosen for their antiandrogenic action, cyproterone acetate, chlormadinone acetate, drospirenone, and dienogest are the most effective.7
Adverse Effects of Oral ContraceptivesThrombosis is one of the most severe adverse effects of OCs and is classically considered to be induce especially by estrogens and their dose. Progestogens, however, also increase the incidence of deep vein thrombosis, especially third-generation gestagens in the first 3 months of use.1 Nevertheless, it should be remembered that the absolute risk of thromboembolism with the use of OCs in healthy patients with no cardiovascular history is low, and is always considerably lower than the risk associated with a natural state such as pregnancy (Table 2). A 2014 meta-analysis by the Cochrane Library found that all oral contraceptives studied (the study included only contraceptives in which the estrogen was ethynylestradiol) were associated with an increased risk of venous thrombosis.6 This risk is similar for OCs with 30–35μg of ethynylestradiol and gestodene, desogestrel, cyproterone acetate, or drospirenone, and in all of them, the risk is higher than 50%–80% in OCs in which the progestogen used is levonorgestrel. The authors therefore consider that the OC of choice (used only for contraceptive purposes) should be that which combines 30μg or less of ethynylestradiol with the levonorgestrel, in women who prefer oral contraceptives.6 That meta-analysis, however, does not include any OCs with natural estrogens, such as estradiol, estrone, or estriol, which have a lower risk of thrombosis than synthetic estrogens such as ethynylestradiol. Moreover, it is important to remember that levonorgestrel applied by means of an intrauterine device has no risk compared to the normal population of venous thrombosis, and yet its efficacy is greater than that of classical OCs. Thus, when dermatologists prescribe contraceptives when there is a need to use potentially teratogenic drugs, we should probably provide information regarding this possibility to our patients.2
Risk of Thromboembolism in.
| Healthy women aged between 15 and 44 years not taking OCs: from 5 to 10 cases per 100,000 women per year |
| Women taking OCs containing less than 50μg of ethynylestradiol with levonorgestrel: 20 cases per 100,000 women per year |
| Women taking OCs containing at least 20μg of ethynylestradiol in combination with desogestrel or gestodene (monophasic, biphasic, or triphasic presentations: 30–40 cases per 100,000 women per year |
| The risk of venous thromboembolism associated with the use of any OC is less than that associated with pregnancy, which is estimated at 60 cases per 100,000 pregnancies |
Another major adverse effect of OCs is hypertension, but this is rare and appears in the first 6 months of use. It appears that women who take OCs and who have no other cardiovascular risk factors (hypertension, smoking) do not have an increased risk of suffering acute myocardial infarction.8 This is despite the fact that OCs have been linked to increased levels of glucose and insulin resistance, and to abnormal lipid metabolism. Synthetic estrogens tend to increase triglyceride levels, whereas natural estrogens do not have this effect. Synthetic estrogens increase triglycerides and HDL and reduce LDL, whereas gestagens have the opposite effect: they increase LDL and reduce HDL. The increased risk of acute myocardial infarction and of acute cerebrovascular accident only arises if OCs are associate with smoking and hypertension.8 A link has also been reported between taking OCs and increased development of hepatic adenoma (3/100,000 users). Other adverse effects are considered to be minor and include nausea and vomiting, weight gain, mastalgia, melasma (rare if OCs with low doses of estrogens are used), acne (androgenic progestogens), and bleeding between periods (more frequent with lower doses of estrogens).
Regarding the influence of the use of OCs on the frequency of cancer, it appears that, overall, OCs play a slightly protective role, as they reduce the incidence of cancer of the uterus, ovaries and colon.9,10 However, OCs appear to slightly increase the risk of breast and cervical cancer. The increase in the relative risk of breast cancer in users of OCs is 1.24 and is limited to users under 40 years of age who began taking the drugs before 20 years of age and maintained treatment for more than 5 years.11,12 Not all the studies corroborated this increased risk of breast cancer due to use of OCs.13 The relative risk of cervical cancer is estimated at between 1.3 and 1.8 in patients who use OCs for more than 5 years; however, this risk exists only in women with persistent human papillomavirus infection and OCs are therefore considered to be cofactors with papillomavirus in the carcinogenesis of cervical cancer.14 Furthermore, some authors deny any pathogenic role of OCs in cervical cancer, as sexually active women are more likely to take OCs and thus more likely to contract human papillomavirus, the etiologic relationship of which with cervical cancer has been unquestionably proven.
Contraindications to Treatment with Oral Contraceptives and Study Prior to IndicationA number of circumstances contraindicate treatment with OCs, almost all of which are linked to a personal or family history of cardiovascular risk factors, but also to any prior condition that contraindicates the use of estrogens (estrogen-dependent tumors). Specifically, we cannot use OCs in any of the situations indicated in Table 3.7
Contraindications of Oral Contraceptives.
| Breastfeeding during the first 6 weeks |
| Smoking more than 15 cigarettes per day in women over 35 years of age |
| Obesity, with a BMI of >40 |
| Multiple associated cardiovascular risk factors (age, smoking, diabetes, hypertension, waist measurement >88cm) |
| Uncontrolled hypertension: systolic above 160mmHg or diastolic above 100mmHg |
| Personal history of deep vein thrombosis or pulmonary embolism |
| Deep vein thrombosis/current pulmonary embolism |
| Major surgery with prolonged immobility |
| Diagnosed familial thrombophilia: situations with thrombogenic gene mutations |
| Ischemic heart disease |
| Ischemic stroke |
| Heart valve disease complicated by pulmonary hypertension, atrial fibrillation, a history of endocarditis, or metal prostheses |
| Diabetes mellitus of more than 20 years or associated with vascular disease, neuropathy, or kidney disease |
| Migraine headaches with aura |
| Migraine without aura in patients over 35 years of age |
| Current history of breast cancer (past 5 years) |
| Active viral hepatitis |
| Decompensated cirrhosis |
| Hepatic adenoma or liver cancer |
Before prescribing an OC, we should therefore take a complete and directed patient history to rule out situations that contraindicate its use. Blood pressure should also be taken before prescribing and the body mass index (BMI), which is the weight divided by the square of the height (kg/m2), should be calculated. In general, use of OCs is not recommended in women with a BMI of 35kg/m2 or greater, due to the high risk of thromboembolism. Pelvic examination, gynecologic ultrasound, or prior laboratory tests are not generally necessary. Laboratory testing should only be requested if risk factors are present: age >35 years, diabetes, hypertension, or excess weight. In these cases, the tests to request are blood count, coagulation, glycemia, liver function, and lipid count (triglycerides, total cholesterol, HDL, and LDL). In patients with a family history of venous thromboembolism in those under 45 years of age or of atypical location, a thrombophilia study should be performed before prescribing OCs.
After prescribing, a follow-up visit should be made after between 3 and 6 months to facilitate adherence to the method and to answer any queries, but no other specific periodic follow-up is necessary. Thus, preventive health care such as cytology, breast examination, or mammograms should be carried out according to the established schedule according to age range, just as should be done with women who do not take hormonal contraceptives.
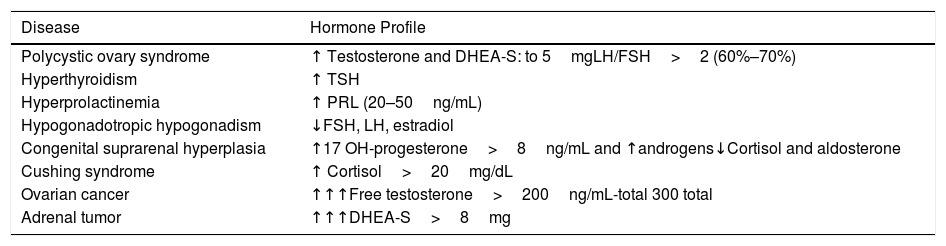
When the indication of the OCs is due to signs of androgenization, a prior hormonal study is indicated and should include the following: testosterone (total and free), DHEA-S (dehydroepiandrostenedione sulfate), 17-hidroxyprogesterone, prolactin, LH and FSH in the first 7 days of the cycle, and cortisol, TSH, and T4 (Table 4).
Simplification of Hormone Profile in Androgenization Syndromes.
| Disease | Hormone Profile |
|---|---|
| Polycystic ovary syndrome | ↑ Testosterone and DHEA-S: to 5mgLH/FSH>2 (60%–70%) |
| Hyperthyroidism | ↑ TSH |
| Hyperprolactinemia | ↑ PRL (20–50ng/mL) |
| Hypogonadotropic hypogonadism | ↓FSH, LH, estradiol |
| Congenital suprarenal hyperplasia | ↑17 OH-progesterone>8ng/mL and ↑androgens↓Cortisol and aldosterone |
| Cushing syndrome | ↑ Cortisol>20mg/dL |
| Ovarian cancer | ↑↑↑Free testosterone>200ng/mL-total 300 total |
| Adrenal tumor | ↑↑↑DHEA-S>8mg |
Modified table of María de la Calle in “El médico de la puerta de al lado”. Madrid, June 2018.
Abbreviations: DHEA-S indicates dehydroepiandrosterone sulfate; LH, luteinizing hormone; FSH, follicle stimulating hormone; TSH, thyroid stimulating hormone; PRL, prolactin.
When using OCs as protection against pregnancy due to teratogenic therapies, the OC of choice, given all of the above, is probably a combination of low doses of ethynylestradiol (20μg) with levonorgestrel. In some women, however, hormonal contraception is contraindicated and, in these cases, other nonhormonal methods may be used.
If the indication for the OCs is to treat signs of androgenization (such as hirsutism, acne, oligomenorrhea, seborrhea, androgenetic alopecia, and infertility), the preferred OCs are those with antiandrogenic progestogens and contraceptives with first-generation and second-generation progestogens, which have a certain androgenic effect, should not be used. The progestogens with an antiandrogenic effect are cyproterone acetate, chlormadinone acetate, drospirenone, dienogest, and nomegestrol acetate; the commercial preparations containing these progestogens are shown in Table 5.
Commercial Preparations with Antiandrogenic Progestogens.
| Cyproterone acetate |
| 2mg cyproterone acetate+35mg ethynylestradiol: Diane® and Gyneplen® |
| 1mg cyproterone acetate+2mg ethynylestradiol: Climen® (hormone replacement therapy) |
| 50mg cyproterone acetate: Androcur® |
| Drospirenone |
| 3mg drospirenone+0.02mg ethynylestradiol: Yasminelle®, Drosurelle®, Dretinelle® |
| 3mg drospirenone+0.03mg ethynylestradiol: Yasmin®, Drosure®, Dretine® |
| 2mg drospirenone+1mg estradiol: Angeliq® (hormone replacement therapy) |
| Chlormadinone acetate |
| 2mg chlormadinone acetate+0.03mg ethynylestradiol: Balianca®, Belara® |
| Dienogest |
| 2mg dienogest+0.03mg ethynylestradiol: Aylin®, Oedien®, Ceciliana®, Danielle®, Donabel® |
| 2mg dienogest+3mg estradiol: Qlaira® |
| Nomegestrol acetate |
| 2.5mg nomegestrol+1.5mg estradiol: Zoely® |
Losing excess weight has been shown to greatly improve the manifestations of polycystic ovary syndrome and the first measure to indicate in these patients, if they are overweight, should be to recommend changes in lifestyle, especially physical activity and a hypocaloric diet.15 The most effective contraceptives in this syndrome continue to be those containing cyproterone acetate or drospirenone, or another antiandrogenic progestogen. OCs are always indicated in premenopausal women with hirsutism and the drugs of choice are therefore those containing antiandrogenic progestogens. If OCs are not sufficient, cyproterone acetate will have to be included in the first 10 days of the cycle or spironolactone throughout the month; a third line may involve associating the OC with finasteride.16 Cyproterone or spironolactone should be used in postmenopausal women with hirsutism. The same course of action may be followed in premenopausal androgenetic alopecia, changing the third line to dutasteride and, in this case, women should avoid pregnancy during treatment and 6 months afterwards if dutasteride is chosen and for a further month thereafter if finasteride is chosen. In postmenopausal women, androgenetic alopecia can be managed with hormone replacement therapy that includes cyproterone acetate as the progestogen, or with finasteride/dutasteride. All OCs appear to improve acne and can be used in women with acne who also want contraception, or when conventional treatments for acne have proven ineffective. One meta-analysis found cyproterone and drospirenone to be the most effective progestogens in the treatment of acne.17 In more specific situations, the OC should be chosen based on the idiosyncrasies of each patient; for example, in patients with weight gain associated with prior use of OCs, a contraceptive with drospirenone may be chosen, which reduces fluid retention because of its good mineralocorticoid profile. The same type of OC (with drospirenone) will also be better accepted in patients who suffer from premenstrual syndrome. In patients with heavy genital bleeding, OCs with dienogest as the progestogen may be chosen, as this molecule reduces the amount and number of days of bleeding. Indeed, a contraceptive with estradiol valerate and dienogest has been approved for the treatment of heavy bleeding. OCs with dienogest may also be chosen in women with irregular bleeding during the cycle, provided they contain more than 20μg of ethynylestradiol, as preparations with low doses of estrogens favor bleeding between periods. Dienogest is also the progestogen of choice in patients with endometriosis. Finally, all OCs have been shown to help improve anemia and dysmenorrhea.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Requena C, Llombart B. Anticonceptivos orales en dermatología. Actas Dermosifiliogr. 2020;111:351–356.