Finegoldia magna is a strictly anaerobic gram-positive coccus that is part of the commensal flora of the skin, oral cavity, gastrointestinal tract, and the female urogenital tract.1,2 We describe an unusual case of a complicated gluteal abscess caused exclusively by an opportunistic pathogen in a patient with diabetes mellitus who had not received previous intramuscular injections in the affected area.
A 60-year-old man came to the emergency department with intense pain and suppuration in the left buttock that had begun 2 weeks earlier. The patient’s past history included hypertension, dyslipidemia, overweight (body mass index, 29), vitiligo, active smoking (20 cigarettes per day), and type 2 diabetes mellitus of 12 years duration with good glycemic control without evidence of metadiabetic complications (HbA1, 7%), although he reported worsening of glycemic control at home in recent weeks, coinciding with the infectious process. The patient reported no previous gluteal intramuscular injections. He was being treated with acetylsalicylic acid (100 mg/24 h), atorvastatin (10 mg/24 h), glimepiride (2 mg/24 h), and bisoprolol (5 mg/24 h).
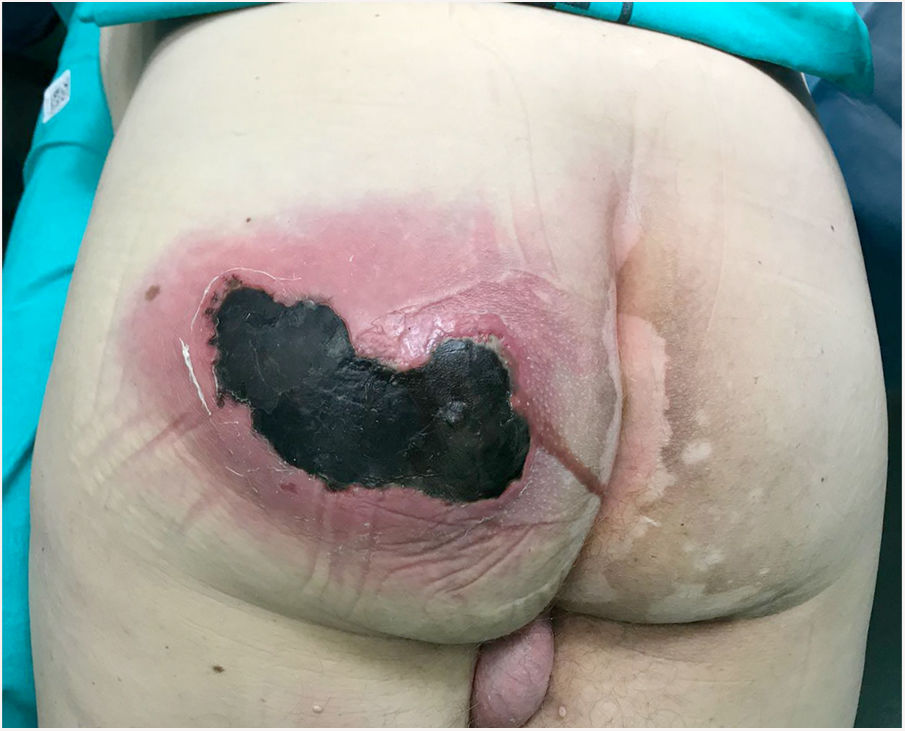
Physical examination revealed a large, painful, hot fluctuant swelling on the left buttock accompanied by crepitus and active suppuration, and a necrotic area of 10 cm (Fig. 1). Laboratory tests revealed the following: hemoglobin, 15 g/dL; leukocytes; 10 870 mm3; platelets, 121 000 mm3; prothrombin time, 16%; glucose, 370 mg/dL; urea, 67 mg/dL; creatinine, 1.25 mg/dL; lactic acid 2 mmol/L; procalcitonin, 1.19 ng/mL; C-reactive protein, 249 mg/L.
Based on the clinical findings the patient underwent emergency drainage in the operating room. A liter of pus was extracted, samples were taken for culture, and wide excision of the necrotic tissue was performed. During the postoperative period the patient showed a favorable clinical course, recovering glycemic control and mobility and functionality of the left leg.
Abscess cultures were negative for aerobic bacteria and positive only for strictly anaerobic gram-positive cocci, which bacterial typing identified as F magna. An antibiogram for F magna revealed sensitivity to amoxicillin-clavulanic acid, metronidazole, and clindamycin, and no evidence of antibiotic resistance. The patient was discharged from hospital and prescribed antibiotic treatment (metronidazole, 500 mg/8 h) and outpatient surgical wound care, to which he responded well.
F magna, previously known as Peptostreptococcus magnus, has undergone multiple taxonomic changes since it was first described in 1933 by Prevot, who used the term Diplococcus magnus.3
F magna is probably the most common of all gram-positive anaerobic cocci (GPAC) and the most pathogenic form found in human clinical samples. This microorganism is part of the normal biota and is found predominantly in the skin, gastrointestinal tract, female genitourinary tract, and, to a lesser extent, the oral cavity.1,2 In recent years, 2 important proteins have been identified that explain the virulence and colonization capacity of F magna: the surface adhesion protein FAF (F magna adhesion factor) and the subtilisin-like serine protease SufA.2,4 In the upper layers of the epidermis, FAF mediates adhesion by binding to galectin-7, a keratinocyte cell marker. After bacteria move deeper into the skin and reach the basal membrane, SufA breaks down collagen IV. In the dermis, FAF interacts with collagen V and fibrillin, which presumably explains how F magna can reach the deepest layers of dermal tissue during infection.5
F magna is one of the species most commonly cultured from abscesses of the skin, soft tissues, bones and joints, usually in polymicrobial cultures, and in rare cases in pure cultures.3 Postoperative mediastinitis, prosthetic valve endocarditis, and necrotizing pneumonia due to F magna have been described.6 The use of broad-spectrum antibiotics that alter the skin microbiota, the introduction of foreign materials (prosthetic valves, replacement joints, catheters), and an increase in the number of patients treated with immunosuppressive drugs favor these infections by creating an optimal environment for these opportunistic pathogens.
Antimicrobial resistance among anaerobic bacteria is increasing worldwide.7 Most GPACs (>90%) are susceptible to penicillin, and many are also susceptible to other β-lactam antibiotics,7–10 but show increasing resistance to other antibiotics such as clindamycin, metronidazole, and fluoroquinolones.8–10 Rates of clindamycin resistance among GPACs range from 7% to 20%, but are increasing in certain species, including F magna and Peptoniphilus species.7 Most GPACs are still susceptible to metronidazole, although some metronidazole-resistant strains of F magna and Parvimonas micra have been described.9
The present case illustrates how the commensal bacterium F magna is capable of producing an advanced necrotizing infection, and underscores the importance of species-level identification and determination of the antimicrobial sensitivity of anaerobic bacteria from representative samples when isolated in pure cultures.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Arencibia-Pérez B, Benet-Muñoz O, Roque-Castellano C, Marchena-Gómez J. Absceso glúteo por finegoldia magna en un paciente diabético. Actas Dermosifiliogr. 2020;111:527–528.