Hydrochlorothiazide and other thiazide diuretics have been used for decades to treat high blood pressure, heart failure, and chronic kidney disease. Thiazides have been linked to photosensitivity with heterogeneous clinical manifestations and recovery times. Diagnosis can be aided by phototesting, photopatch testing, and skin biopsy. Long-term use of hydrochlorothiazide has been linked to an increased dose-dependent risk of certain types of skin cancer in recent years. In this review, we also look at other less common or lesser-known adverse effects of thiazide diuretics that have been described in isolated reports.
La hidroclorotiazida (HCTZ) y otros diuréticos tiazídicos son fármacos que se han empleado desde hace décadas para el tratamiento de la hipertensión arterial, la insuficiencia cardíaca o la enfermedad renal crónica. Las tiazidas se han asociado con reacciones de fotosensibilidad, siendo heterogéneas en cuanto a manifestación clínica y tiempo de recuperación, y en las cuales, el fototest, el fotoparche y la biopsia cutánea nos pueden ser útiles en el diagnóstico. En relación con estos fármacos, en los últimos años se ha evidenciado también un mayor riesgo dosis-dependiente de desarrollar determinados tipos de cáncer cutáneo en pacientes tratados de forma crónica con hidroclorotiazida. En esta revisión se comentan, asimismo, otros efectos adversos menos habituales o reconocidos de los diuréticos tiazídicos reportados de forma aislada en la literatura.
Thiazide diuretics began to be used in clinical practice in 1957. These drugs are classified into 2 groups according to their structure: thiazides and thiazide-like agents. For more than 50 years, thiazides and thiazide-like agents have been a cornerstone in the treatment of hypertension, both in monotherapy and in combination with other antihypertensive agents, as the effects are additive in terms of reducing blood pressure.1 They have also been widely used in situations of cardiac overload such as heart failure and chronic kidney disease (CKD).2
Mechanism of actionThiazide diuretics act by blocking the sodium chloride (NaCl) channel in the proximal segment of the distal convoluted tubule. On blocking the NaCl channel, lower levels of sodium pass through the luminal membrane, thus decreasing the action of the sodium/potassium (Na/K) pump and limiting the passage of Na+ and water to the interstitial space.3
The method of activation of thiazide diuretics is to induce a change in Na+ concentration distal to the distal convoluted tubule. Subsequently, the ion channels and pumps work to equilibrate the imbalance in Na+ levels. Blocking the NaCl channel leads to an increase in sodium and water retention in the lumen and a decrease in Na+ in the distal convoluted tubule. At the same time, blocking the NaCl channel increases ion flow through the NaCa channel, thereby leading to increased reabsorption of calcium in the interstitial space in exchange for return of Na+ to the distal convoluted tubule. Through aldosterone-mediated sodium retention that triggers an increase in Na+ flow to the collecting duct system, there is an increase in Na+ resorption and excretion of K+ and H+ ions in urine.4
PharmacokineticsThiazide-like diuretics are quickly absorbed in the gastrointestinal tract. Food intake increases absorption of hydrochlorothiazide (HCTZ), whereas heart failure or CKD lowers absorption. Thiazides bind strongly to plasma proteins for distribution, and so glomerular filtration is limited and the agents are excreted mainly via the kidneys, although there are marked differences in metabolism and excretion5,6 (Table 1).
Pharmacokinetic characteristics of the main thiazide diuretics.
| Thiazide diuretic | Bioavailability, % | Peak, h | Volume of distribution, L/kg | Effect duration, h | Half life, h | Percentage renal excretion |
|---|---|---|---|---|---|---|
| Hydrochlorothiazide | 70 | 4–6 | 0.83 | 6–12 | 6–14 | 95 |
| Chlorthalidone | 65 | 2–6 | 0.14 | 40–72 | 47 | 65 |
| Indapamide | 95 | 1–4 | 24 | 11 | 70 | |
| Metolazone | 65 | 2–4 | 1–1.5 | 24–48 | 24–48 | 80 |
| Xipamide | 95 | 1–2 | 12–20 | 5–8 | 30 |
Chlorthalidone has a much longer half-life (40–72h) and a greater volume of distribution than HCTZ because 98% of the drug binds to carbonic anhydrase in erythrocytes, reaching concentrations 7–10 times greater than in plasma. Therefore, red blood cells act as a reservoir from which the drug is released to plasma and gradually eliminated via tubules in renal excretion. Indapamide and metolazone also bind to carbonic anhydrase in erythrocytes, have a large volume of distribution, and a longer half-life than HCTZ. Thiazides are not efficacious in patients with severe CKD because the glomerular filtration rate (GFR) limits the load of filtered Na+ reaching the distal tubule, and at this site the efficacy is modest (Table 2).
Cutaneous adverse effects reported secondary to treatment with diuretic thiazides, mainly hydrochlorothiazide.
| Thiazide diuretics | |
|---|---|
| Photosensitivity8–17 | Most reactions present in the form of sunburn-like lesionsHeterogeneous clinical presentation requiring thiazides to be suspected as the causal agentPersistent photosensitivity can be observed in some patients, despite discontinuation of the drug |
| Skin cancer18–28 | Increased risk of dose-dependent SCC and possibly MCC and malignant adnexal neoplasmsNo increased risk or low risk of BCC or MMDiscrepancy regarding CTCL |
| Pruritus and ezcema29–32 | Approximately 1% of patients developed chronic pruritusGreater frequency of eczematous processes, whether photodistributed or not |
| Drug-induced lupus33–38 | Essentially cutaneous forms: SCLE above all |
| Autoimmune bullous diseases39,40 | Doubtful relationship with development of BPNo cases described of pemphigus vulgaris |
| Drug-induced lichen planus and lichenoid rash41–45 | Possibility of DRL, particularly in photoexposed areas |
| Miscellaneous conditions | Small-vessel vasculitis16,46,47AGEP48,49 |
Abbreviations: AGEP, acute generalized exanthematous pustulosis; BCC, basal cell carcinoma; BP, bullous pemphigoid; CTCL, cutaneous T cell lymphoma; DLR, drug-induced lichenoid rash; SCC, squamous cell carcinoma; MCC, Merkel cell carcinoma; MM, malignant melanoma; SCLE, subacute cutaneous lupus erythematosus.
Thiazide diuretics do not reduce blood pressure in patients with normal blood pressure and are only efficacious in those with hypertension. The initial decrease in blood pressure is associated with inhibition of the NaCl channel, as explained above, which increases diuresis and natriuresis, decreasing plasma volume and reducing venous return and cardiac output.7
As with other antihypertensive agents used in monotherapy, only 40%–60% of those treated with thiazide diuretics achieve acceptable control of blood pressure. The main predictors of response include higher baseline blood pressure, female sex, shorter duration with diagnosed or treated hypertension, low levels of active plasma renin, and greater decrease in urinary excretion of sodium. Negative predictive factors, in contrast, include mainly the patient's age.7
AdministrationThiazide diuretics are administered orally in the form of tablets. Patients should take their tablets in the morning with food. Generally, for treatment of hypertension, a low dose (25mg/day) is required; this can be increased to 50 or 100mg, depending on the individual therapeutic needs of the patient. In patients who suffer a greater cardiac overload, dosing can start at 50–100mg with switching to 50–200mg, depending on the agent used.
Thiazides and photosensitivityApproximately 8% of adverse drug reactions take the form of photosensitive rash.8 In the article published by Blakely et al.9 in 2019 on drug-induced photosensitivity, HCTZ is considered to belong to the most common group of photosensitizers, with 60 cases published in the literature. Although urticaria seems to be the most frequent form of drug-induced cutaneous reaction, other less common forms have been reported such as symmetrical drug-related intertriginous and flexural exanthema (SDRIFE).10
The pathophysiological mechanism to explain why thiazides act as photosensitizers has yet to be determined. In vitro studies have shown direct damage to DNA11 and cell membrane lipids.12
The clinical presentation comprises the development of a symmetrical rash on photoexposed areas (face, neck, anterior trunk, back of the hands, and forearms). The onset of lesions can occur with differing degrees of severity and with different clinical patterns13:
- •
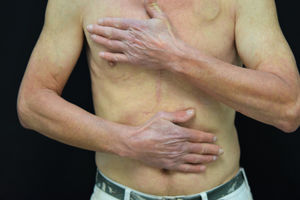
Sunburn-like rash (Fig. 1). The lesions are mainly comprised of well-defined erythema, with variable edema, which is normally associated with pruritus and/or stinging sensation. The severity of the rash varies from one patient to another. This type of reaction appears more frequently in black individuals, regardless of the duration of treatment.
- •
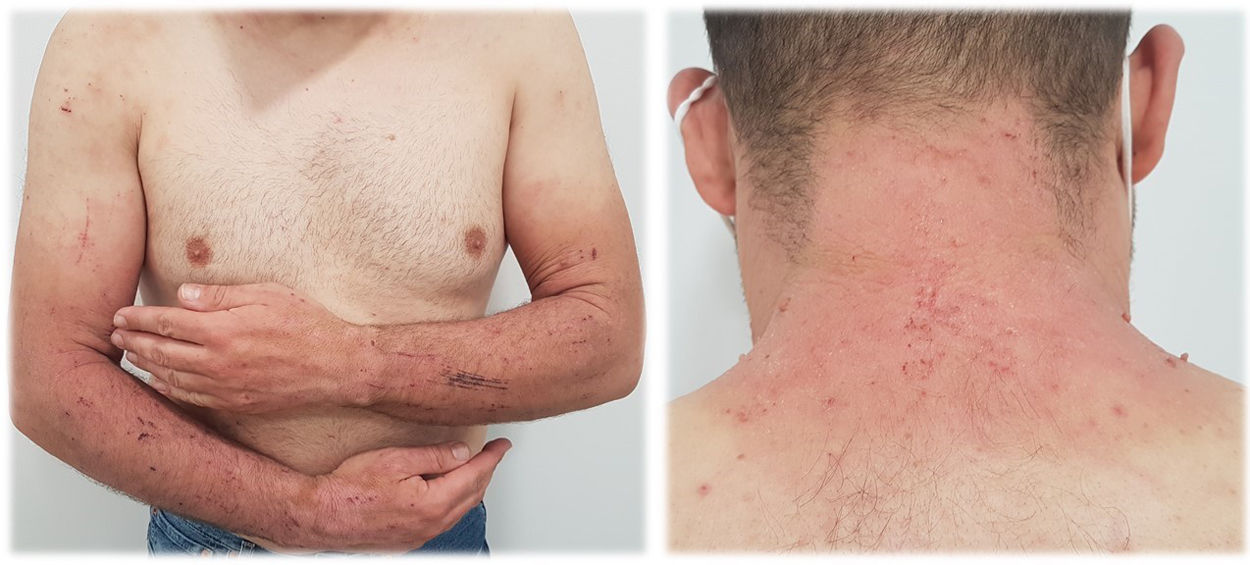
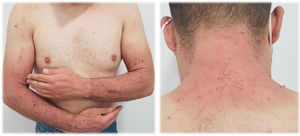
Eczema-like rash (Fig. 2). These lesions correspond to an acute or subacute dermatitis of variable severity, normally itchy, with erythema progressing rapidly to peeling. In these cases, skin patch testing is almost mandatory.
- •
Subacute lupus-like rash. This is indistinguishable from idiopathic subacute cutaneous lupus, both in clinical and histopathological terms. Some patients present antinuclear antibodies and anti-Ro/SS-A and/or anti-La/SS-B antibodies.
- •
Miscellaneous conditions. There have been reports of multiple forms of thiazide-induced photosensitive rash, such as lichenoid lesions, cheilitis, pseudoporphyria, petechiae, photo-onycholysis, photoleukomelanoderma, and even persistent photosensitivity conditions.
It is clear that the clinical presentation of photosensitivity is heterogeneous, such that clinical suspicion should be high in order to arrive at a correct diagnosis. A review of the drugs taken by the patient and the temporal relationship between thiazide intake, changes in dose, and onset of rash should help establish suspicion of a relationship with the drug. According to data published by Tsai et al.,14 most patients developed HCTZ-induced photosensitive rashes in the first 3 years of treatment, regardless of age and/or sex. Although not available in all clinics, a photobiological study of these patients is recommended to determine the causal relationship between the drug and the photosensitive rash.
By means of phototesting, it is possible to determine which part of the radiation spectrum is responsible for the photosensitive rash. First and foremost, the minimal erythema dose (MED) for ultraviolet B (UVB) and response to UVA should be studied. The most common situation is the presence of photoreactions to UVA, followed closely by reactions to UVA+UVB. In exceptional cases, photoreaction to visible light may be associated with reaction to UVA and UVB, and still less frequently, to UVB alone. The article published by Selvaag15 considers a possible synergistic effect between UVA and UVB radiation in the development of thiazide photosensitivity reactions. Ideally, phototesting should be performed while the patient is still taking the thiazide, and subsequently treatment discontinued before repeating phototesting to assess objective changes in the determination of the reaction to different parts of the light spectrum. However, this is not always possible and the patient usually discontinues the drug in accordance with the treating physician's indications before assessment in a photobiology clinic.
The time to resolution or normalization of photoreactions varies from one patient to another, although photosensitivity is expected to resolve in the first 6 months. It is therefore recommended to repeat phototesting 3–4 weeks after discontinuing the drug, and in the event that photosensitivity persists, repeat phototesting every 2–3 months until the test results are normal (increase of 40% or more in MED measured while taking the drug). In cases in which photosensitivity persists beyond 6 months (some clinics wait for 12 months), the possibility of an etiology other than one based on thiazides should be considered (although we should not forget that there is a persistent form photosensitivity associated with this drug, although this is infrequent). In addition to that discussed above, HCTZ increases the risk of hypersensitivity reactions to alopurinol when administered concomitantly. This is another of the drugs implicated in drug-induced cutaneous reactions, although not principally in the form of photosensitivity.
It is recommended to perform a biopsy of the rash to determine the histological pattern, and this will enable a differential diagnosis to be made in the patient. A case of HCTZ-induced vasculitis was reported,16 in which biopsy was key; a mast cell degranulation study was subsequently performed.
The photopatch should also be considered as a complementary test in these patients, although the diagnostic yield of the test is lower compared with the phototest. The allergen HCTZ 10% petroleum jelly can be obtained as a patch test product for drug-induced cutaneous reactions (Chemotechnique Diagnostics®). Even though this drug is considered as one of the main causes of photosensitivity reactions, it does not form part of the photopatch battery in any of the main distributors so commercial allergen can be used for the photopatch (it should be patched in duplicate, occluded for 48h, and then one of the sites should be irradiated with UVA 5J/cm2, whereas the other should not be irradiated). A reading at 96h is essential for interpretation of the result. The patient should be reminded to use dark clothing during the week of the study to avoid bias in the results. Although the test is usually negative, given it is widely available (more so than a photobiology test), its use is recommended. Positivity in the test exclusively at the UVA-irradiated site confirms a photoallergic reaction to the drug, whereas if both sites (the irradiated and nonirradiated one) are positive, it is considered a photo-aggravated contact dermatitis to thiazide. It should be remembered that a negative study does not rule out the possibility that a patient may experience a photosensitive rash caused by the drug.
Management of these patients is apparently simple, although at times the outcome is not the desired one when the drug is interrupted. The first decision will be to assess whether it is possible to discontinue treatment (both in cases when the aforementioned complementary tests have confirmed the causal agent and when there is an unconfirmed suspicion that the agent is responsible for the photosensitive rash). In most cases, it will be possible to discontinue treatment or switch to another drug in a different class or one with less sensitizing potential (such as furosemide or indapamide); in the event that this is not possible, rash severity should be assessed along with occupational and social activities associated with exposure to sunlight. The use of the drug at night could potentially decrease the photosensitizing potential; however, the use of diuretics at night may have a significant impact on the patient's rest, making it an unviable option. The use of sunscreens (mainly physical/mineral) and the use of dark clothing can help avoid or reduce skin effects. At times, the patient may report an impact on quality of life associated with symptoms such as pruritus. These may be managed with oral antihistamine agents and/or corticosteroids (topical or oral), with variable responses, usually unsatisfactory in most cases. Robinson et al.17 considered the possibility of phototherapy treatment, similar to hardening used with solar urticaria. However, despite the success reported in the article, further studies strengthening the evidence with this therapeutic option have not been published. Given the cross-reactions with para-phenylenediamine, or para-aminobenzoic acid derivatives, the progress of the patient could be compromised on indicating sunscreens that contain the latter allergen; skin patch tests with photopatches could be of use to study such patients.
Skin cancerNonmelanoma skin cancer (NMSC) is the most common neoplasm in individuals with light skin types and its incidence is increasing.18 In recent years, there is a growing interest in the relationship between NMSC and treatment with thiazide diuretics, particularly in the case of HCTZ. Epidemiological studies performed in Denmark in 2018 based on data from national databases found that, with a cumulative HCTZ dose of 50000mg, the relative risk (RR) was 1.3 and 3.9 for basal cell carcinoma (BCC) and squamous cell carcinoma (SCC), respectively, compared with the unexposed population or the population exposed to lower doses.19 The risk therefore appears to be dose-dependent, as it increases in patients with cumulative HCTZ doses in excess of 200000mg, with a RR of 1.54 for BCC and 7.38 for SCC.19 The meta-analysis by Shin et al.20 and Bendinelli et al.21 arrived at similar results, with a more prominent relationship between HCTZ use and SCC risk compared with BCC. A population-based study in individuals aged over 65 years further supported the dose-dependent increase in risk of overall NMSC in thiazide-treated patients, but not other classes of antihypertensive agents.22 The photosensitizing activity of thiazides could explain this increase in NMSC incidence through increased risk of actinic damage.18
With regards malignant melanoma (MM), as for BCC, the risk appears minimal or non-existent, but there are some contradictions in the literature. As above, a retrospective analysis of national databases showed an increase in MM in these patients, particularly the lentigo maligna and nodular melanoma subtypes.23 However, a recent article with a sample size of more than 20000 patients did not find any relationship between HCTZ use and increased risk of MM or BCC, unlike for SCC, in which the risk increased with increasing duration of treatment and cumulative dose.24 Recently, a cohort study was published to assess the possible risk in patients who initiated treatment with HCTZ or other thiazide-like agents, once again showing an increased risk of SCC in HCTZ-treated patients, but no increased risk for BCC or MM.25 Indapamide and bendroflumethiazide were also studied; these agents have been associated with a greater incidence of MM, but no significant association was found for any type of skin cancer.
There also appears to be an increased dose-dependent risk of Merkel cell carcinoma (MCC) and malignant adnexal neoplasms with use of HCTZ,26 although further studies have not been published. Finally, the relationship between HCTZ and cutaneous T-cell lymphomas (CTCLs), essentially mycosis fungoides, is not clear: Jahan-Tigh et al.27 observed a higher prevalence of HCTZ use in patients diagnosed with CTCL compared with the general population, as well as a possible temporal relationship, and proposed that HCTZ could be acting as an antigen trigger for MF in a small subset of patients. After discontinuing the drug, the MF lesions disappeared in several patients, whereas rechallenge with HCTZ led to their reappearance or exacerbation. Nevertheless, a recently published article reached the opposite conclusion, suggesting that chronic exposure to HCTZ could act as a protective factor for development of CTCL.28
Pruritus and ezcemaDrugs are a recognized cause of pruritus, particularly in elderly and polymedicated patients.29 Of particular note seems to be the relationship between pruritus and drugs used for cardiovascular diseases, with the pruritus rate in HCTZ-treated patients close to 1%; this is similar to the rates associated with angiotensin converting enzyme (ACE) inhibitors, beta blockers, amiodarone, and statins.30 Whereas pruritus associated with ACE inhibitors may occur as a result of increased bradykinin levels, the underlying mechanism for HCTZ-induced pruritus is thought to be cutaneous inflammation.30 Thiazides are a cause of eczematous reactions, whether or not associated with photosensitivity, and a statistically significant relationship has been established between chronic eczema in patients treated with thiazides compared with the control group.31 It has been postulated that HCTZ could affect skin barrier function given its mechanism of action involves blockade of the NaCl cotransporter and that sodium channels play a role in epidermal differentiation.32
Drug-induced lupusDrug-induced lupus erythematosus (DILE) is an autoimmune disorder similar to lupus that usually appears with chronic exposure to certain drugs and that resolves after discontinuation of the culprit agent.33 There is a growing list of drugs implicated in inducing forms of cutaneous lupus erythematosus; these may be both subacute (SCLE) or chronic.34 Thiazide diuretics have essentially been associated with cutaneous forms, which generally present clinically as erythematous scaly plaques and are histologically indistinguishable from an idiopathic SCLE. In addition, the presence of anti-Ro/SS-A and anti-La/SS-B antibodies can be detected in serum, as well as deposition of immunocomplexes in the basement membrane.13 The first case of DILE attributed to HCTZ was reported in the scientific literature in 1985.35 Since then, new cases have emerged. Cases have also been linked to the combination of HCTZ with triamtherene36 and to other thiazides such as chlorothiazide.37 Finally, a case of systemic lupus erythematosus (SLE) has also been reported associated with diuretic thiazides.38
Autoimmune bullous diseasesDifferent drugs have been implicated as causal agents of bullous pemphigoid (BP).39 Drug-induced BP can be an acute and self-limiting disease that resolves after discontinuing the culprit agent or a chronic process that appears to be triggered by the drug and follows the course of a classic form. Currently, there are few case reports of HCTZ as a possible causal agent in which an improvement occurs in lesions or they resolve completely in these patients after discontinuing the diuretic.39.40 Studies have not been found that link thiazide diuretics with pemphigus vulgaris.
Drug-induced lichen planus and lichenoid rashDrug-induced lichenoid rashes (DLR) present as papules or scaly plaques with eczematous or psoriasiform characteristics, distributed symmetrically with a predilection for the trunk and limbs; these rashes are often distributed on photoexposed sites. Unlike DLR, lichen planus tends to be more localized, more frequently affecting flexural sites, mucosa, and genitals, and presents Wickham striae, which are usually absent in DLR.41 The cases of DLR described associated with thiazide diuretics are essentially photodistributed conditions that resolve rapidly after discontinuation of HCTZ.42,43 Sin et al.44 reported a case of HCTZ-induced lichen planus without exposure to sunlight, with the association considered probable according to the Naranjo algorithm. Likewise, chronic and generalized DLR has been reported in a middle-aged woman with chronic lymphatic leukemia; the rash was attributed to an oral combination of irbesartan and HCTZ.45
Miscellaneous conditionsAlthough very infrequent, HCTZ can be responsible for cutaneous small-vessel vasculitis (CSVV).16,46,47 In addition, to date, cases of acute generalized exanthematous pustulosis (AGEP) have been reported.48,49 Hypersensitivity reactions due to cross-reactivity are possible in patients allergic to sulfonamides.50
Conflicts of interestThe authors declare that they have no conflicts of interest.