Common warts are viral infections caused by the human papillomavirus. They can occasionally represent a therapeutic challenge, particularly at certain sites such as the subungual or periungual regions and on the soles of the feet. Although there is an extensive therapeutic arsenal for the treatment of these warts, resistance to treatment is common at these sites.
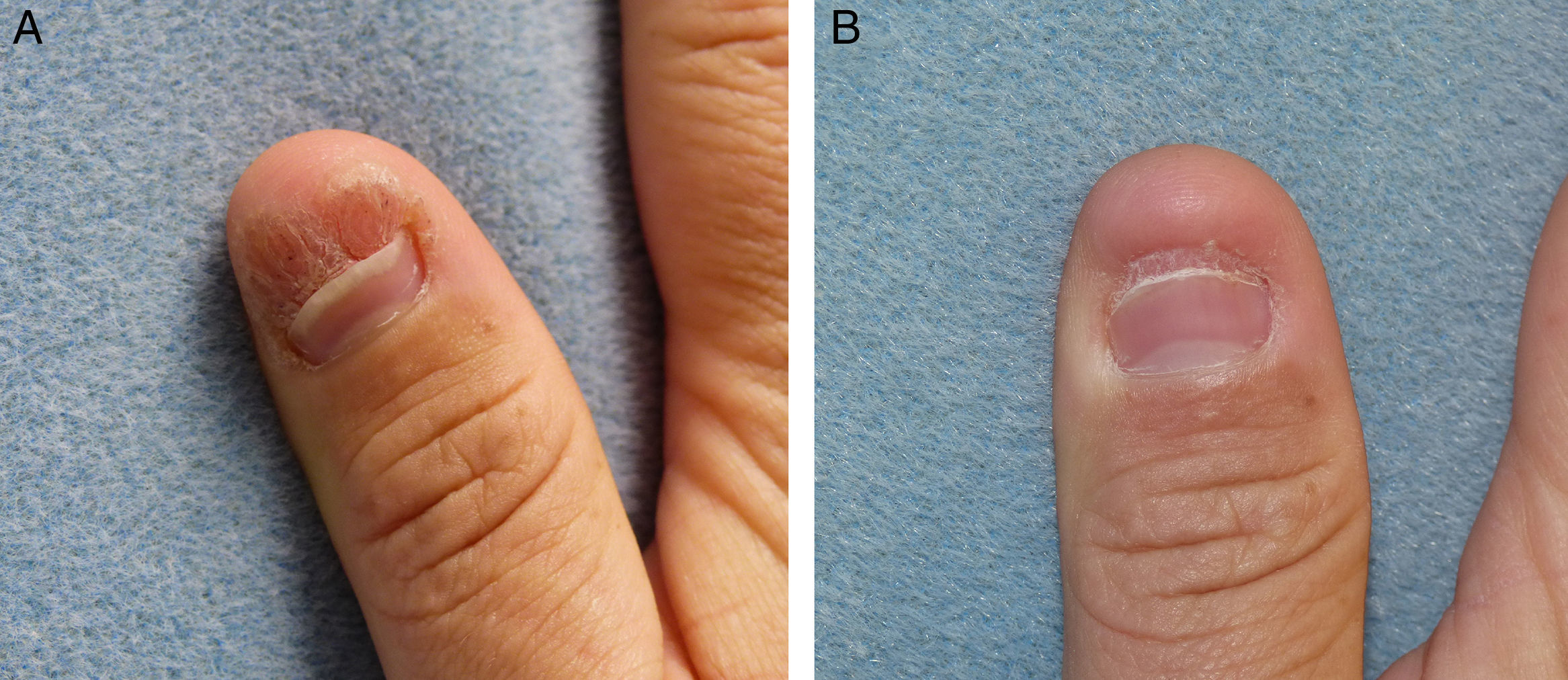
A 45-year-old man, with no past medical history of interest, was seen for a subungual wart that had been present on the first digit of the right hand for 2 years, causing nail dystrophy, with pain and limitation for performing domestic and occupational tasks that required use of the finger-thumb pincer grip (Fig. 1A). Numerous treatments, summarized in Table 1, had been attempted, including in combination, but had been unsuccessful. After performing biopsy to rule out verrucous carcinoma, it was decided to start immunotherapy treatment with the trivalent mumps, measles, and rubella (MMR) vaccine by local injection. Injections of 0.3ml (each vial contains 0.5ml) were performed at 2-week intervals. No previous immunization was required as the patient had received MMR vaccine in childhood. After 5 sessions, inflammatory changes were observed in the area, with the appearance of edema and erythema, and after the seventh session the wart disappeared completely (Fig. 1B). No adverse effects were observed, except for moderate pain during the injection, and there has been no recurrence after more than a year of follow-up.
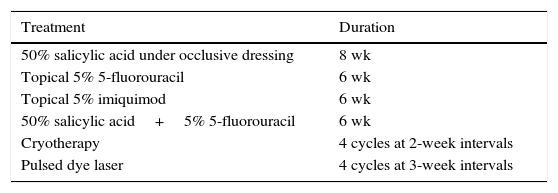
Previous Treatments in Our Patient and Their Duration.
| Treatment | Duration |
|---|---|
| 50% salicylic acid under occlusive dressing | 8 wk |
| Topical 5% 5-fluorouracil | 6 wk |
| Topical 5% imiquimod | 6 wk |
| 50% salicylic acid+5% 5-fluorouracil | 6 wk |
| Cryotherapy | 4 cycles at 2-week intervals |
| Pulsed dye laser | 4 cycles at 3-week intervals |
The treatment of common warts at certain sites, such as the subungual region, can be frustrating. A wide range of therapies with different mechanisms of action have been tried, most commonly involving destructive methods or immunotherapy.1 Immunotherapy techniques include contact allergens, such as diphencyprone and dinitrochlorobenzene, and immunomodulators, which can be topical, such as imiquimod, or oral, such as cimetidine and zinc sulfate.2 In addition, ever more trials of intralesional immunotherapy are being performed, with antigens such as candidin and the tuberculosis antigen, and with vaccines such as the mumps or MMR vaccines.3 Although the mechanism of action remains unclear, this therapy is thought to induce a Th1-cell–mediated immune response. Cytokines released in the response include interleukin-2 and γ-interferon, which are released in much higher concentrations at the site of injection, focusing the local immune response in that area and favoring elimination of the human papillomavirus.4 Local trauma may contribute to intensification of the response. Furthermore, the immunomodulatory effect may act not only locally, but also at a distance, as it is not uncommon to see the disappearance of warts at distant sites.5 For this immune response to develop, the patient must already have been exposed to one or more of these antigens, either by previous vaccination in accordance with the vaccination calendar, or by direct contact with the virus in the past.5 When patients have not been immunized, an initial dose of subcutaneous vaccine is recommended. The complete response rate varies between 27% and 81%, with each patient requiring a mean of 3.6 cycles at 2-week intervals.3 The most common adverse effects include pseudoinfluenza symptoms and pain at the site of injection; no serious adverse effects have been observed.3 Advantages of intralesional over traditional topical immunotherapy include its cost, the availability of antigens, and possible effects at a distance from the injection site.4 The vaccine can be obtained from the hospital pharmacy or directly from a high-street pharmacy with a medical prescription. The cost of a vial of vaccine is of approximately 15 euros, which may be subsidized by the health service provider.
In conclusion, the availability, cost-effectiveness, and absence of serious adverse effects of intralesional immunotherapy with the trivalent vaccine make it a useful option for the management of warts refractory to other treatments.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Morales-Raya C, Maroñas-Jiménez L, Aragón-Miguel R, Postigo-Llorente C. Mumps, Measles, and Rubella Vaccine for the Treatment of a Recalcitrant Subungual Wart. Actas Dermosifiliogr. 2017;108:966–968.