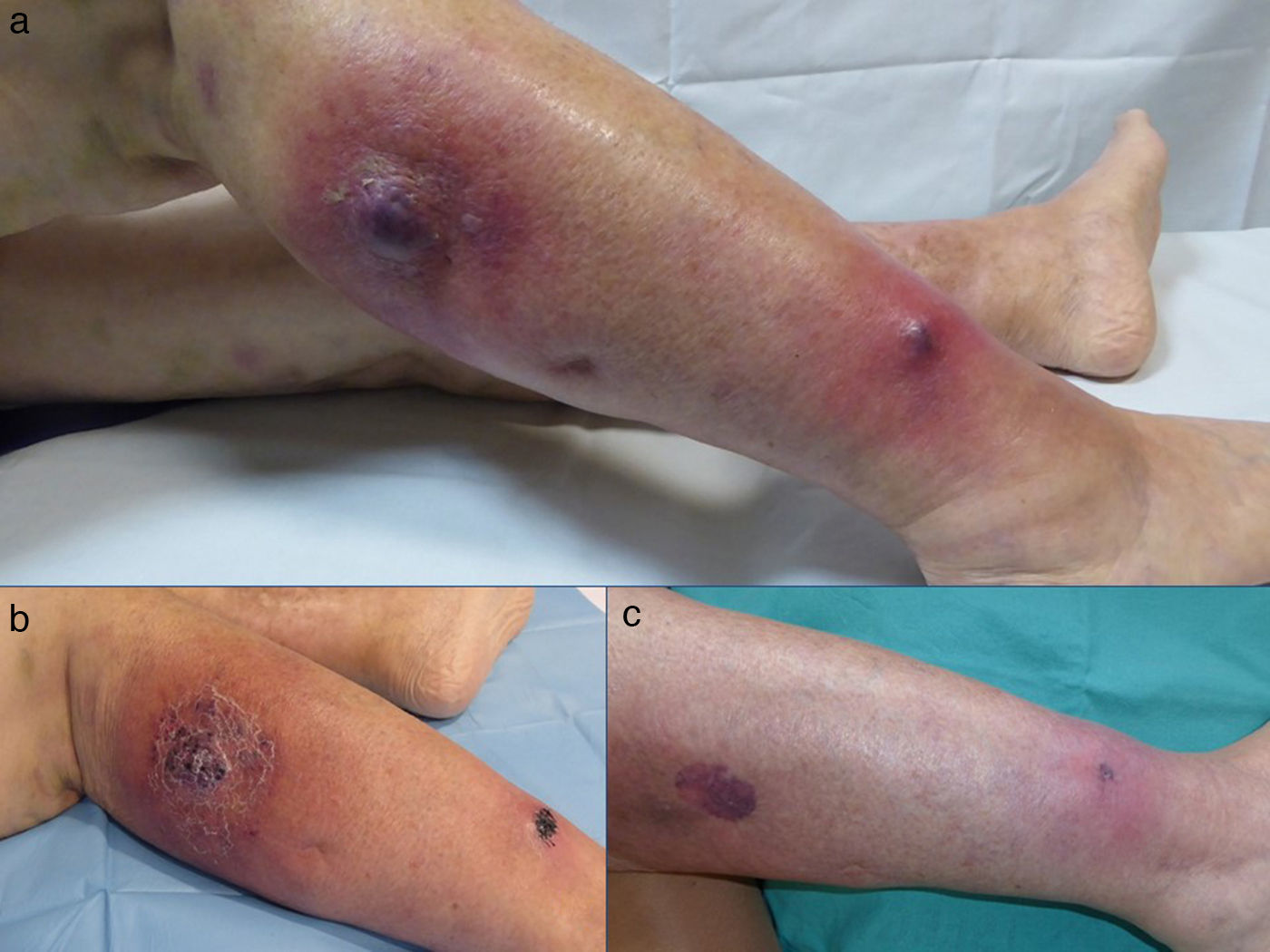
A 78-year-old woman, with a past history of systemic hypertension and a chronic myeloproliferative syndrome, came to the dermatology emergency room for the presence of 2 painful, erythematous-purpuric lesions that had arisen on her left lower limb a week earlier. She stated that the lesions were increasing in size and that the pain now interfered with walking. She did not report fever or other associated symptoms. Dermatologic examination revealed 2 poorly defined, tender erythematous-violaceous nodules on her left leg (Fig. 1A).
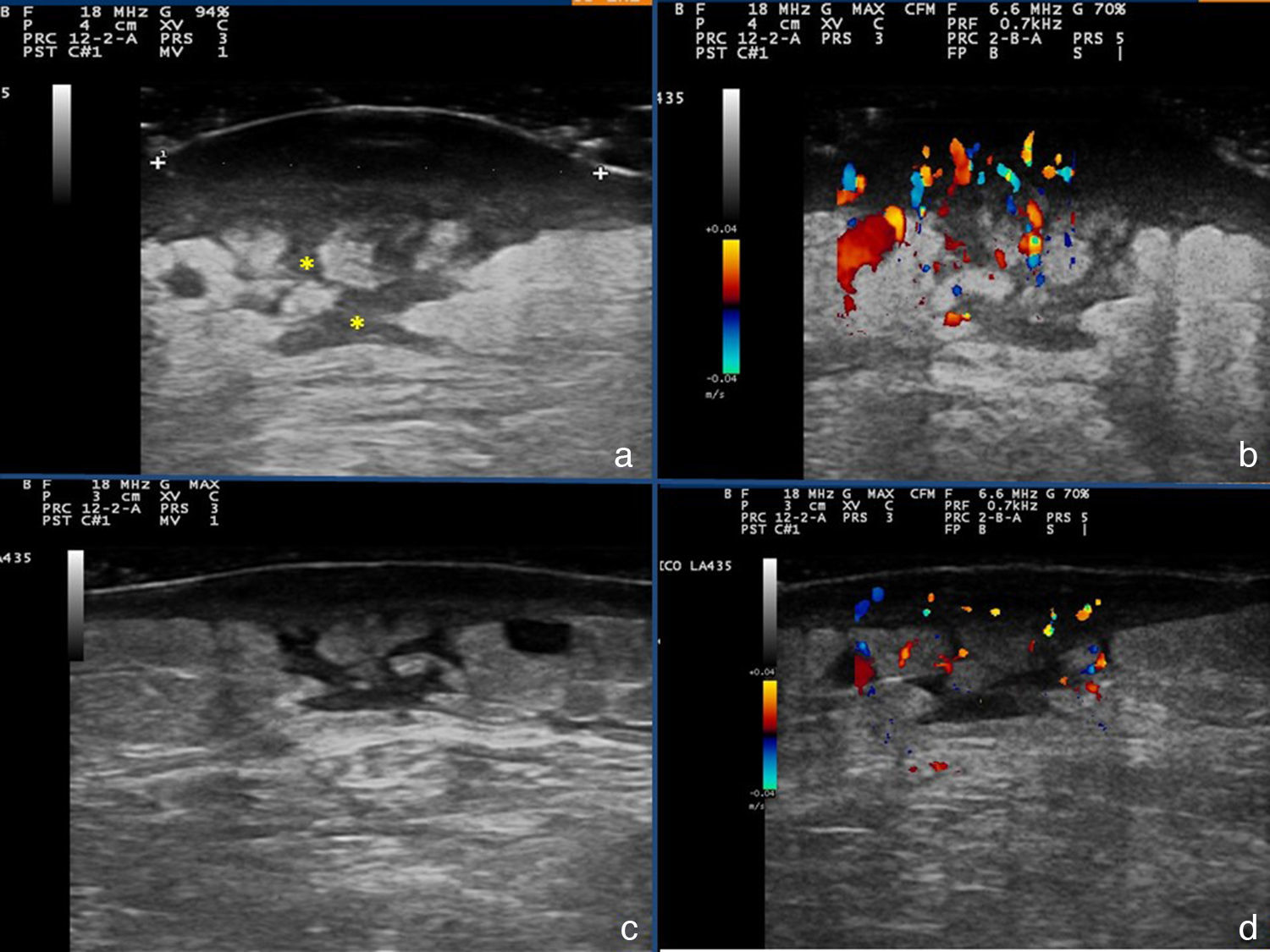
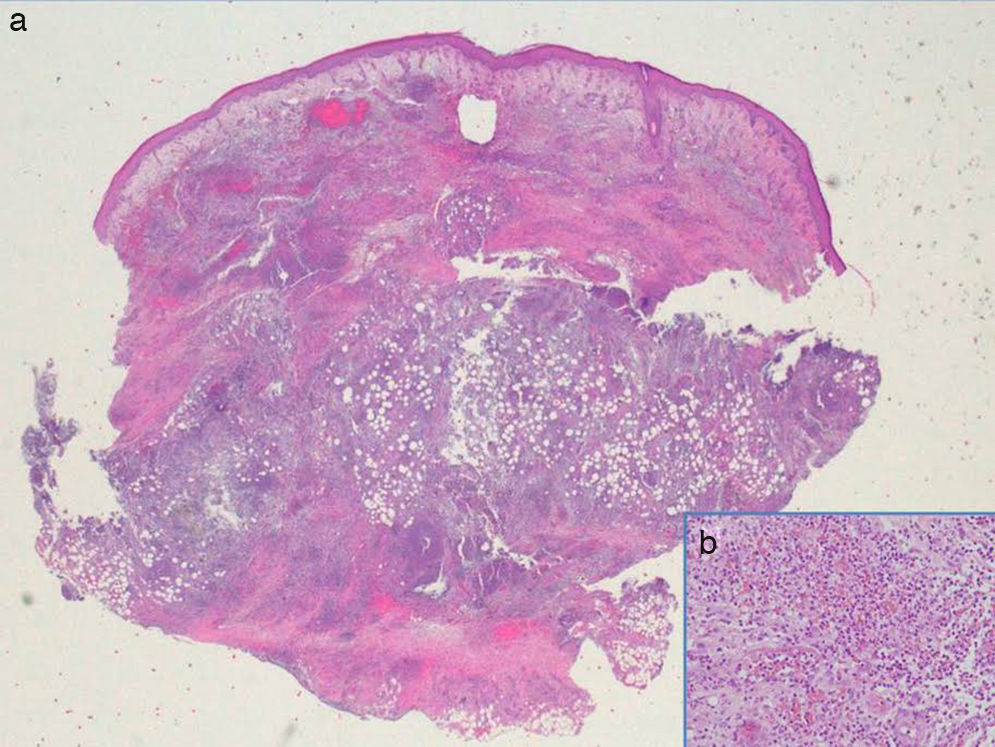
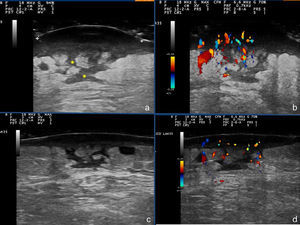
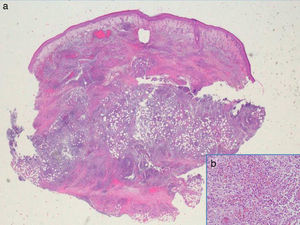
Additional tests included skin ultrasound with a linear 18MHz probe (Esaote MylabGold 25), which revealed a well-defined, hypoechoic subepidermal structure measuring 3.29×1.14cm. This area was continuous with an irregular, heterogeneous hypoechoic area that formed tracts that extended into a destructured hypodermis. Color Doppler showed increased local vascularity. Other additional tests included a complete blood count, which showed no evidence of infection, cultures, which were negative, and a skin biopsy from one of the lesions, which revealed an abundant neutrophilic inflammatory infiltrate in the dermis and that extended into the subcutaneous cellular tissue, as well as foci of abscess formation in the dermis, dissecting through the tissue (Fig. 3). Based on these findings, we made a diagnosis of chronic myeloproliferative syndrome-associated pyoderma gangrenosum (PG) and started treatment with oral prednisone at a dose of 1mg/kg/d.
The patient's clinical follow-up (Fig. 1,B andC) included serial ultrasound studies. After a week of treatment, ultrasound showed a reduction in local vascularity and a decrease in the size of the lesion to 2.87×0.88cm (Fig. 2,A-D); we therefore started to taper the treatment.
Skin ultrasound: A and B, Initial phase. The asterisks mark linear hypoechoic areas corresponding fistulous tracts. C and D, Image after a week of treatment: thinning of the hypoechoic subepidermal band, and decreased vascularity in the color Doppler image. The hypoechoic tracts persist in the subepidermal tissue.
Clinically, PG presents as a painful papule, pustule, or nodule that progresses to a violaceous ulcer with undermined borders.1 Fistulous tracts draining into this ulcer can be observed on ultrasound. The correlation between the ultrasound and pathology findings helps to confirm the clinical diagnosis. Four clinical variants of PG have been described: ulcerative or classic, bullous, pustular, and vegetative or superficial granulomatous.2
PG is associated with a systemic disease in 50% of cases, the most common being inflammatory bowel disease, followed by arthritis, hematologic neoplasms, and autoinflammatory syndromes.3 The etiology and pathogenesis of PG are unknown and diagnosis is based on a compatible medical history and histopathological examination and the exclusion of other diseases that can present similar findings. Different diagnostic criteria have been proposed for PG, but the exclusion of other similar diseases is always an important consideration, with ultrasound study being particularly important for this purpose.4 The differential diagnosis should include infections, nodular syndromes, vascular ulcers, and Sweet syndrome.4
Soft tissue infections such as cellulitis and abscesses can be identified on ultrasound. In cellulitis, there is extensive destructuring of the hypodermis and the image tends to be hyperechoic, in contrast to abscesses, in which the collection of pus is observed as a hypoechoic or anechoic area with no color Doppler signal. In our patient we observed a dermo-hypodermal lesion with an intralesional Doppler signal.5,6
The treatment of the PG is based on immunosuppressive drugs such as topical or systemic corticosteroids, depending on clinical severity and on associated comorbid conditions. In cases like ours, systemic corticosteroids (prednisone, 1mg/kg/d) administered by mouth are the treatment of choice. This achieves a rapid control of disease progression, and therapy can then be tapered in parallel with the clinical signs.7 We followed the clinical course on ultrasound, observing a reduction in the hypoechoic subepidermal band and the hypoechoic hypodermal tracts, as well as restructuring of the subcutaneous cellular tissue and deep dermis. In parallel with these findings, there is a reduction in vascularity visible on color Doppler imaging.
In conclusion, we have described a case of pyoderma gangrenosum with its ultrasound findings as a novel feature in the literature, demonstrating the usefulness of this imaging study for follow-up of the clinical course of this disease. However, further studies are needed to establish an ultrasound pattern and the value of ultrasound in the follow-up of PG.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Pousa-Martínez M, Sánchez-Aguilar D, Aliste C, Vázquez-Veiga H. Utilidad de la ecografía en el diagnóstico y seguimiento del pioderma gangrenoso. Actas Dermosifiliogr. 2017;108:962–964.