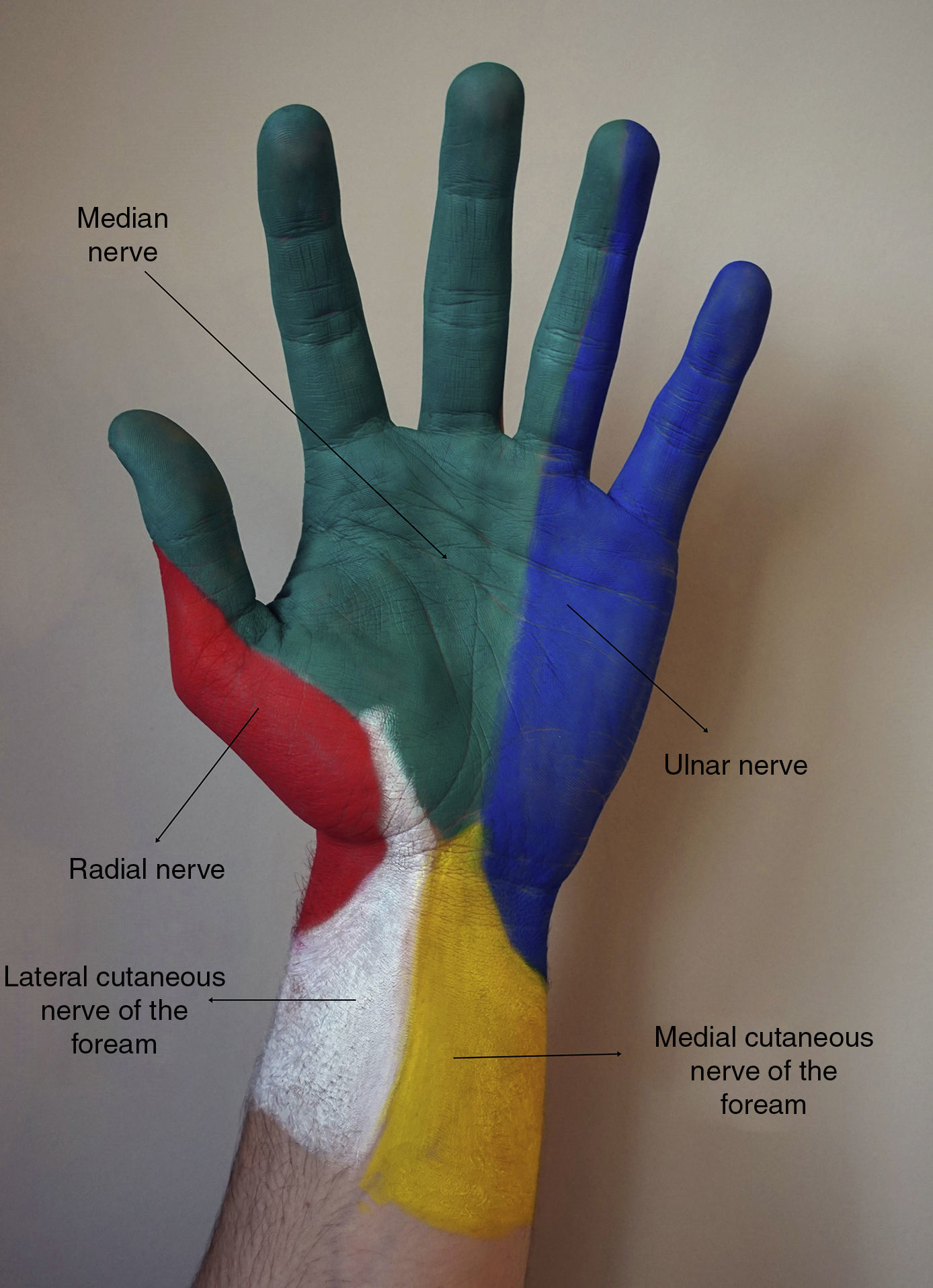
Botulinum toxin (BT) injection is an effective and widely used technique for the treatment of palmar hyperhidrosis (PH). However, the high density of nerve endings in the palmar skin makes pain during the procedure a limiting factor, and topical anesthesia is often insufficient. It is therefore essential to perform an effective anesthetic technique that will relieve pain during the procedure. Sensory innervation to the hand is from superficial branches of the median, radial and ulnar nerves (Fig. 1). These peripheral nerves can be blockade at different anatomical sites, with the wrist being the preferred area as it has the most reliable anatomical references and here the nerves are mainly formed of sensory branches.1
Peripheral nerve blockade with the subsequent injection of BT carries certain risks, notably neuropathy and muscle weakness. To perform the procedure with a minimal risk of complications requires appropriate knowledge of the anatomical structures involved.
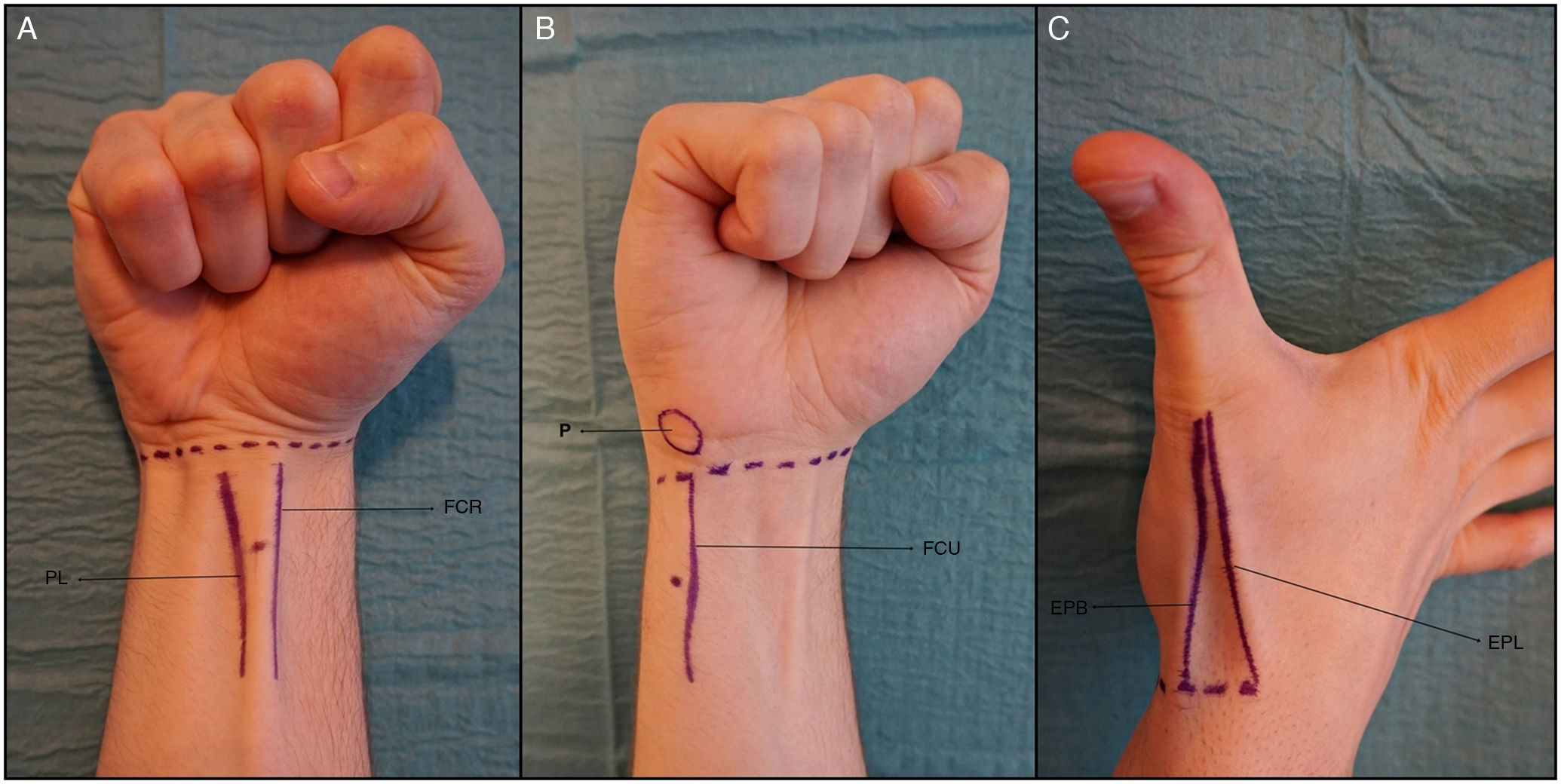
Description of the TechniqueAfter applying antiseptic to the area to be treated, we perform peripheral nerve blocks of the median, ulnar, and radial nerves. The median nerve runs deep in the sulcus formed between the tendons of flexor carpi radialis and palmaris longus. With the wrist held in slight flexion to tense both tendons, the point of injection is located between the tendons, 3cm proximal to the distal wrist crease (Fig. 2A).
Peripheral nerve blocks at the wrist. A, Median nerve block. B, Ulnar nerve block. C, Radial nerve block.
Abbreviations EPB, extensor pollicis brevis tendon; EPL, extensor pollicis longus tendon; FCR, Flexor carpi radialis tendon; FCU, flexor carpi ulnaris tendon; P, pisiform bone; PL, palmaris longus tendon.
For ulnar nerve block, the most useful anatomical references are the pisiform bone and flexor carpi ulnaris tendon. The point of injection is situated immediately deep to the flexor carpi ulnaris tendon, 2cm proximal to the pisiform bone (Fig. 2B). Before injecting the anesthetic solution, it is important to aspirate to ensure that the tip of the needle is not in the ulnar artery. For each nerve (median and ulnar), the injection is of 2mL of 2%mepivacaine, using sterile 30G needles (Sterican, Braun, Melsungen, Germany).
Finally, for radial nerve block, the thumb in placed in extension to identify the extensor pollicis longus tendon medially and the extensor pollicis brevis tendon laterally. These 2 tendons border the anatomical snuffbox, into which the anesthetic agent is widely infiltrated (Fig. 2C).
A vial of 100 units of BT type A (Botox, Allergan, Irvine, CA) is reconstituted using 5ml of sterile 0.9% saline solution. Between 75 and 100 units are typically used in each palm, depending of the size of the hand. The injections are performed intradermally, using sterile 30G needles, separating the points of injection by 1 to 1.5cm. Two units (0.1mL) are injected at each point. Very superficial injections of lower doses are recommended over the thenar eminence in order to minimize the risk of muscle weakness secondary to diffusion of the toxin into the underlying muscles.
The onset of the effect of treatment begins at 48 to 72hours and the mean duration of efficacy is of 6 to 8 months. However, recent studies have shown a significant increase in the duration of efficacy of treatment with BT for PH after successive treatments.2
IndicationsBT is approved for the treatment of severe and persistent axillary hyperhidrosis that interferes with the activities of daily living; its use in the treatment of PH is off-label. However, there is extensive scientific evidence supporting its good efficacy and safety both in adults and in children.3
ContraindicationsBT is contraindicated in pregnant or breast-feeding women. Likewise, special care must be taken in patients with neuromuscular diseases or who are on treatment with drugs that could interfere with neuromuscular activity (for example, aminoglycosides). The toxin is of course contraindicated in patients with known hypersensitivity to BT or to any of the excipients of the formulation.4
ComplicationsPeripheral nerve blockade at the wrist is a safe procedure when performed using a correct technique, but it is not risk-free. Direct injection into a nerve trunk must be avoided to minimize the risk of neuropathy; if the patient describes paresthesia during the procedure, the needle should be withdrawn slightly and the direction corrected by changing the angle of insertion a few degrees.5
An interesting study found no differences in anesthetic efficacy when shorter needles (30G×6mm) were used to perform the peripheral nerve blocks, compared with conventional, larger diameter needles, reducing the risk of complications.6
As with any injection, the procedure can cause erythema, infection, or localized hemorrhage or hematomas.
ConclusionsBT infiltration for the treatment of PH is a very effective procedure, in which the main limiting factor is pain during treatment. However, adequate knowledge of the regional anatomy and adherence to a series of recommendations makes this a safe procedure with a low rate of complications and high level of satisfaction in the majority of the patients.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
We would like to thank Dr. Xandra Piris García for her inestimable help in producing this video.
Please cite this article as: de Quintana-Sancho A, Calvo MC. Tratamiento de la hiperhidrosis palmar con toxina botulínica mediante bloqueo de los nervios periféricos al nivel de la muñeca. Actas Dermosifiliogr. 2017;108:947–949.