The patient was a 54-year-old man who had received a lung transplant for treatment of emphysema. He was being treated with tacrolimus, mycophenolate mofetil, and prednisone. Rapidly progressive asymptomatic erythematous-violaceous plaques appeared on the patient's chest at 7 months posttransplant. The patient did not present fever or any other associated signs or symptoms.
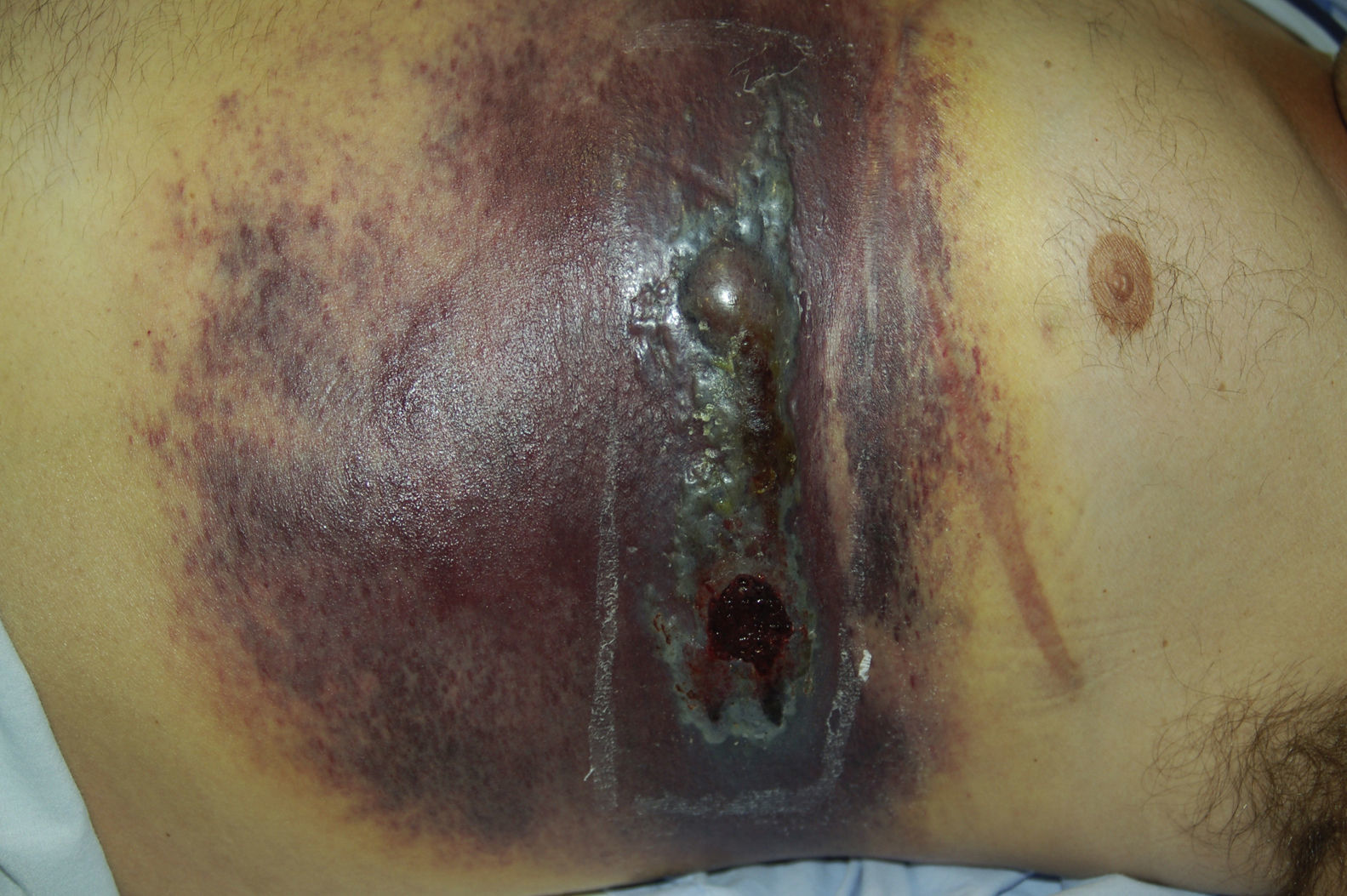
Physical ExaminationAt the first visit, physical examination revealed isolated infiltrated erythematous plaques with a bilateral distribution in the pectoral region (adjacent to the thoracotomy site). One month later, the patient presented a large plaque that covered the lower and lateral parts of the chest and abdomen with 2 tumors, 1 of which was necrotic, on the surface (Fig. 1).
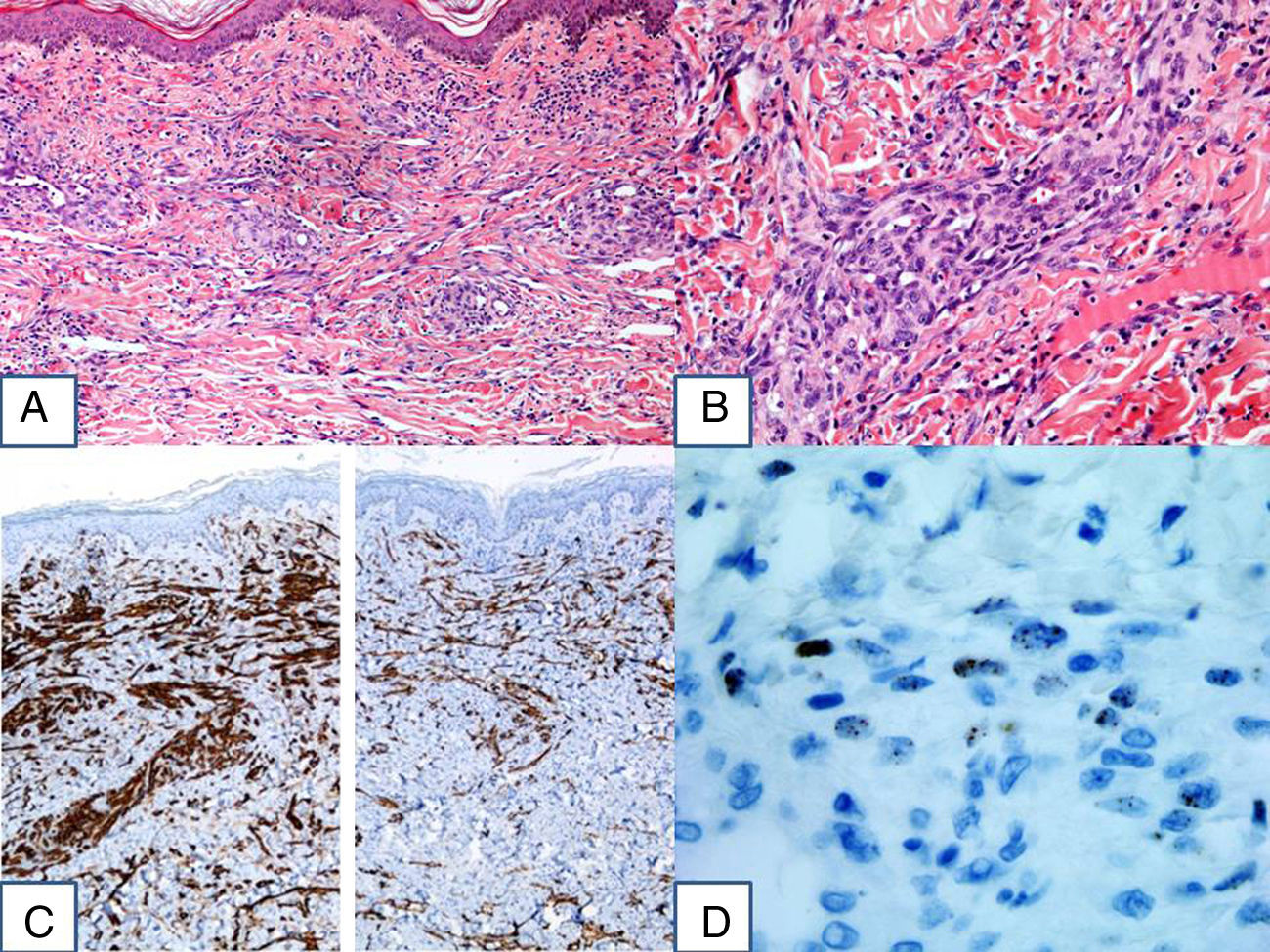
HistopathologyBiopsy revealed a proliferation of spindle-shaped cells throughout the full thickness of the dermis that formed pseudovascular clefts and fascicles. The cells showed moderate atypia and occasional mitotic figures. Immunohistochemistry was positive for CD31, D2-40, and human herpesvirus 8 (HHV8) (Fig. 2).
What Is Your Diagnosis?
DiagnosisIatrogenic Kaposi sarcoma.
Clinical CourseAt the time of the transplant, serology for HHV8 was positive in the donor and negative in the recipient. The patient presented seroconversion for HHV8. Subsequently, HHV8 was detected by polymerase chain reaction (PCR) in the serum of the patient, with high levels of viremia (>1000copies/mL).
Computed tomography and magnetic resonance imaging studies were carried out and no evidence of visceral involvement was detected.
Treatment was initiated with doxorubicin, mycophenolate was withdrawn, the dose of tacrolimus was decreased, and everolimus was introduced. With this treatment regimen, new lesions stopped appearing and the existing lesions flattened.
CommentKaposi sarcoma is a tumor derived from endothelial cells. It presents a variable clinical course, ranging from minimal involvement to systemic progression that affects internal organs. There are 4 types of Kaposi sarcoma: classical, endemic, iatrogenic, and human immunodeficiency virus (HIV)–associated.1
Kaposi sarcoma develops in 0.1%-5% of solid organ transplant recipients, especially kidney recipients, who generally receive combined immunosuppressive therapy.1
Immunity and viral infection play a role in the etiology and pathogenesis of Kaposi sarcoma and interact with HHV8, which is a necessary but not sufficient condition for the development of the disease.2
Most cases in transplant patients are caused by the reactivation of a previous infection, while a few are caused by a primary infection related to the transplanted organ.3
Approximately 30% of seronegative recipients seroconvert after receiving an organ from a HHV8-seropositive donor. Lebbe et al.4 monitored 454 HHV8 seronegative transplant recipients for 2 years and observed a seroconversion rate of 29%-31%. Notably, the prevalence of HHV8-positive viremia in these patients was very low, <1% (4 of 454 patients), possibly due to an adaptive immune response against HHV8 demonstrated by Lambert et al.5 Of the 4 patients with HHV8-positive viremia, 2 had high levels of viremia (>1000copies/mL) and developed Kaposi sarcoma.
The authors concluded that HHV8 transmission is common and that morbidity caused by the virus is rare but can be severe. Therefore, they recommend screening donors for HHV8 in cases in which the recipient is seronegative.
The study did not include any patients who, like our patient, had received a lung transplant (possibly because kidney transplant is the most common solid organ transplant) and although we have not found any cases of Kaposi sarcoma in lung transplant recipients in the reviewed literature, it can be assumed that the situation is similar given that these patients also receive combined immunosuppressive therapy.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Salgüero Fernández I, Alfageme Roldán F, Suarez Massa D, Roustan Gullón G. Placas infiltradas de rápida evolución en paciente trasplantado. Actas Dermosifiliogr. 2017;108:255–256.