The patient was a 37-year-old woman with no history of interest who consulted for a lesion on her right cheek. The lesion was asymptomatic and first appeared more than 5 years previously. It had grown rapidly during the last 3 months.
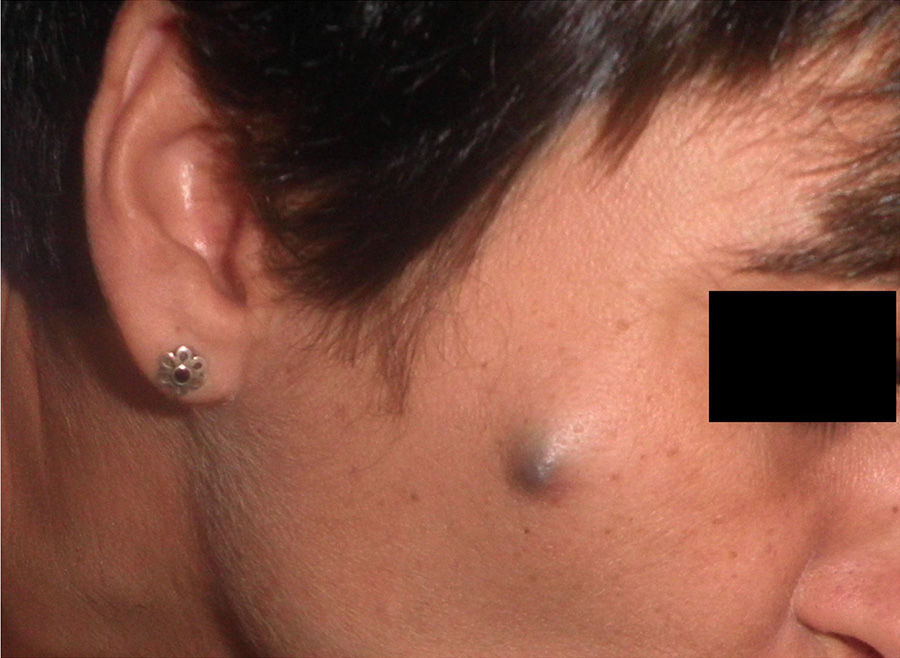
Physical ExaminationPhysical examination revealed a poorly defined nodule measuring approximately 1cm in diameter. The lesion was bluish-gray in color, with no surface ulceration or desquamation, and was attached to the deeper layers (Fig. 1). A complete examination of the skin and mucosa revealed no similar lesions, and no enlarged nodes were found in accessible areas.
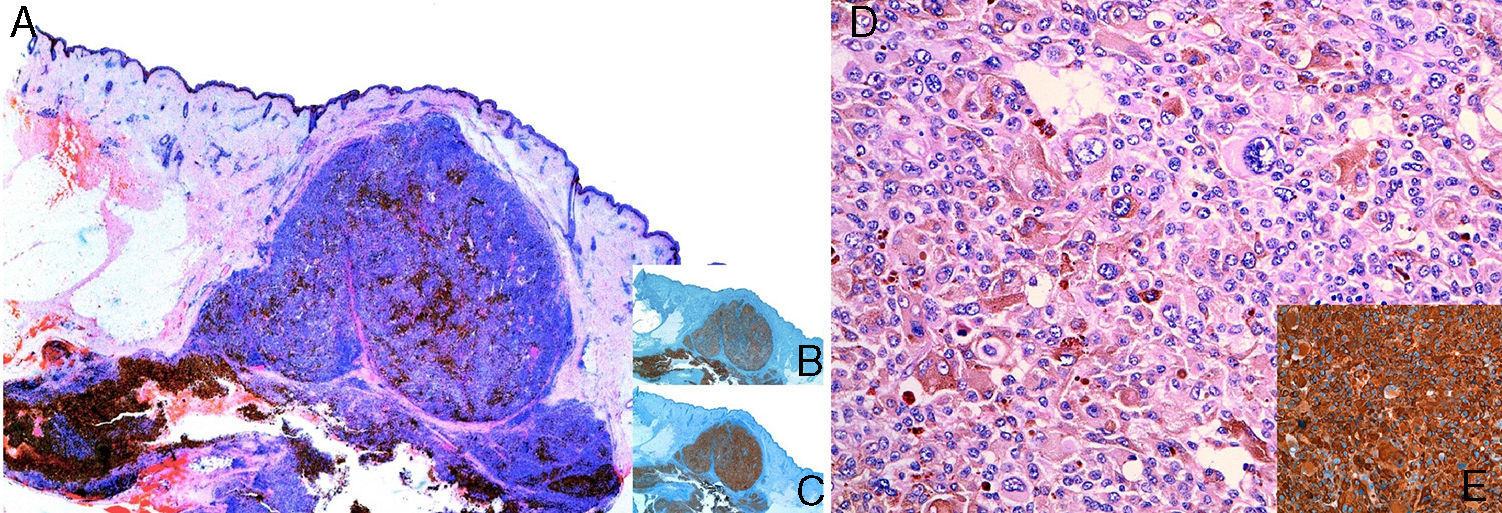
HistopathologyThe lesion was completely removed, and histopathology revealed a multinodular skin lesion that was well defined with cellular atypia, mitosis, and areas of necrosis. The lesion was not connected to the follicular epithelium or epidermis (Figs. 2A and D). Immunohistochemistry was positive in the neoplastic cells for Melan-A, S100, and HMB45 (Figs. 2B, C, and E) and negative for cytokeratins. The study was completed with p53 (95% positive), cyclin D1 (31%), Ki-67 (44%), and D2-40 (low).
Additional TestsThe extension study comprised positron emission tomography, otorhinolaryngologic and ophthalmic examinations, and computed tomography of the neck, chest, abdomen, and pelvis. No pathologic findings were detected.
What is your diagnosis?
DiagnosisPrimary dermal melanoma.
Clinical Course and TreatmentThe Breslow thickness was 13mm. We extended the margin for study by 2cm and performed a study of the sentinel node, which was negative. The patient was then treated with high-dose adjuvant interferon for 1 year. She remains disease-free 3 years later.
CommentPrimary dermal melanoma is a subtype of melanoma that accounts for <1% of malignant melanocytic neoplasms. Its survival rate is higher than might be expected compared with other malignant melanocytic neoplasms with a similar Breslow thickness.1
In 2000, Bowen et al.2 coined the term primary dermal melanoma to refer to dermal melanocytic tumors and tumors of the subcutaneous cellular tissue that fulfilled a series of characteristics, such as location in the dermis, absence of a junctional component or connection with the follicular epithelium, absence of regression and necrosis, and absence of preexisting melanocytic nevus.
In their series published in 2008, Cassarino et al.1 performed an immunohistochemistry study including p53, cyclin D1, Ki-67, and D2-40 in patients with primary dermal melanoma and found statistically significant differences in the expression of these markers compared with patients diagnosed with metastatic melanoma and nodular melanoma. The authors suggested that low levels of expression of genes associated with oncogenesis (defective p53), the cell cycle, and proliferation (cyclin D1 and Ki-67) could be associated with less aggressive primary dermal melanoma. Furthermore, expression of D2-40, which is related to tumor lymphangiogenesis, has been associated with tumor lymphatic invasion and reduced survival.3 Although these findings are not diagnostic criteria in themselves, they could prove useful in the differential diagnosis with other malignant melanocytic neoplasms.1
There have been reports of individual cases and small series of patients with primary dermal melanoma.1,4,5 In the Spanish medical literature, we found only 1 published case.6 In the present case, we performed an immunohistochemistry study using the panel proposed by Cassarino et al.1 Although levels of expression of cyclin D1 and D2-40 were lower than those observed in metastatic melanoma, we recorded high levels of expression of p53 and Ki-67.
The overall survival rate at 5 years for patients with a single cutaneous metastasis of melanoma is 5%-19%, with a mean survival of 7 to 15 months.1,6 In their series of patients with primary dermal melanoma, Swetter et al4 reported a 5-year survival rate of 100%, which clearly differs from that expected in cases of metastatic melanoma and primary cutaneous melanoma with a similar Breslow thickness.4
The differences in the prognosis and therapeutic management of primary dermal melanoma and metastatic melanoma have led the former to be considered an independent entity, whose diagnosis depends on an appropriate clinicopathologic correlation and an exhaustive extension study. Therefore, primary dermal melanoma should be taken into consideration in patients with melanoma confined to the dermis and no evidence of primary lesions at other levels.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Espinosa Lara P, Pérez González Y, Jiménez Reyes J. Nódulo asintomático facial. Actas Dermosifiliogr. 2017;108:59–60.