Morphea and lichen sclerosus et atrophicus (LSA) are 2 chronic skin diseases of unknown etiology and pathogenesis. A possible relationship with genetic, autoimmune, and infectious factors has been proposed. Very few cases have been reported after the administration of vaccines.
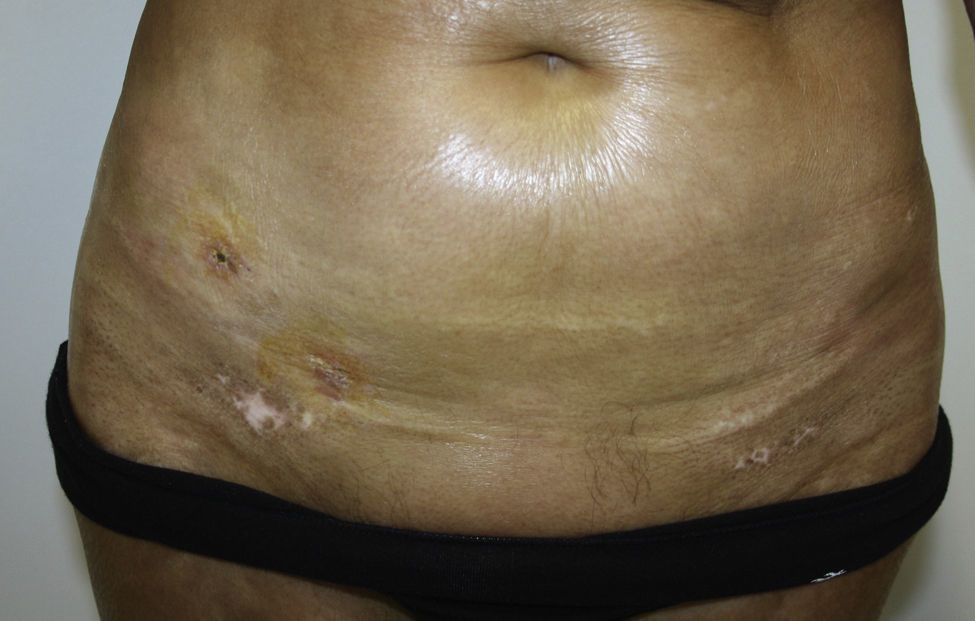
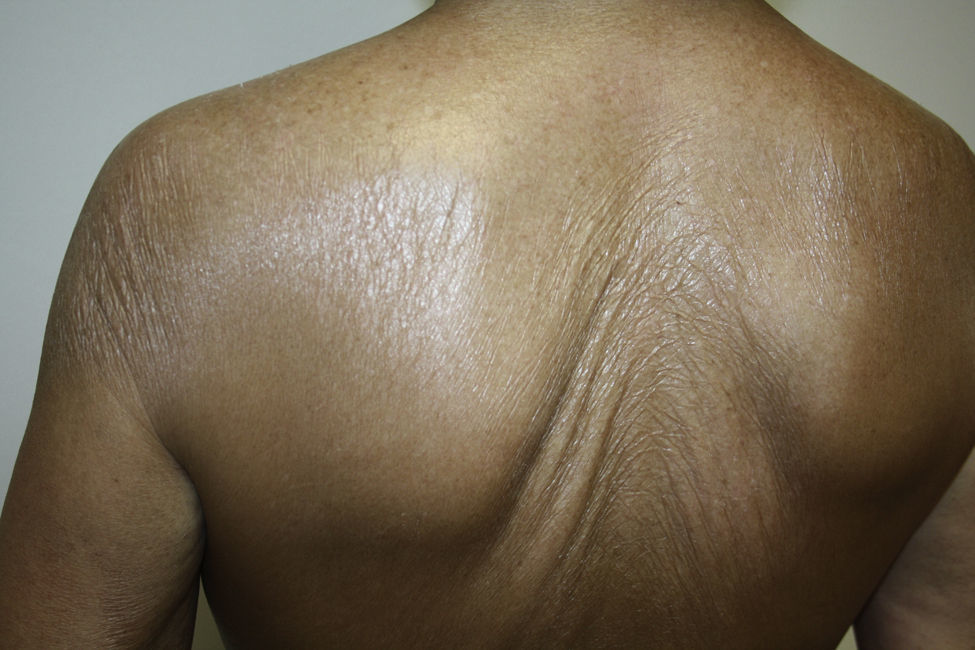
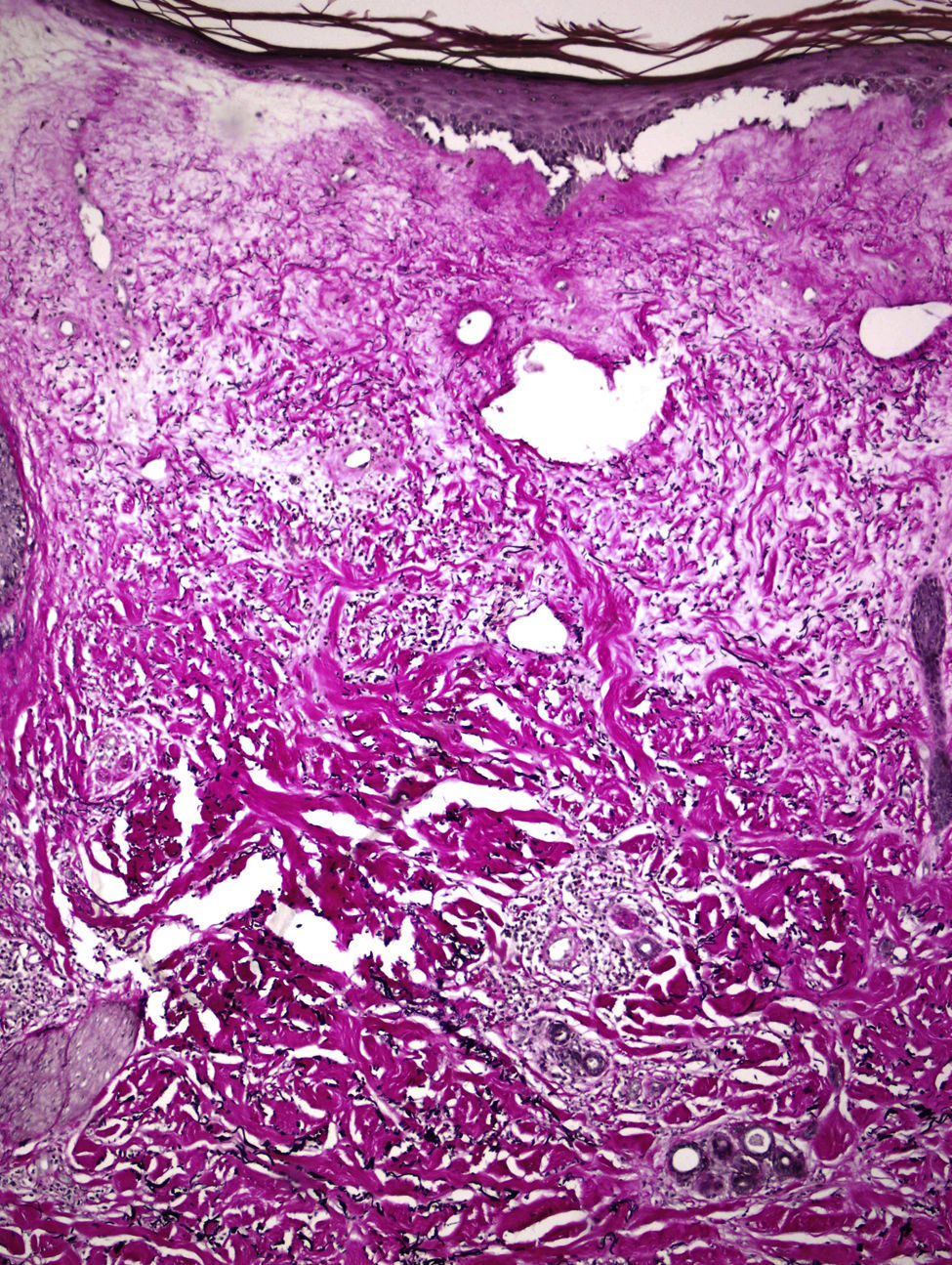
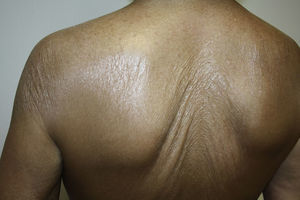
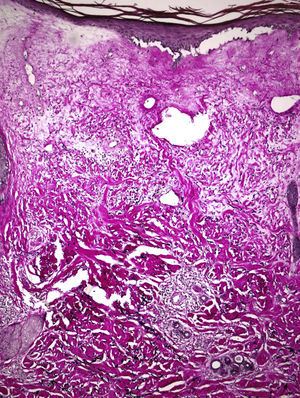
A 67-year-old woman with a past history of breast cancer treated by radical mastectomy and implantation of a silicone prosthesis 15 years earlier, was referred to our department for pruritus and progressive induration of the skin. She had not received radiotherapy after her mastectomy and was taking no drugs of interest. She reported the onset of symptoms 10 days after having received the first dose of 0.5ml of Chiroflu, an influenza vaccine of inactivated surface antigen, administered intramuscularly into her left deltoid muscle. The lesion was initially limited to her left deltoid region, the site of injection of the vaccine. However, generalized lesions gradually developed, respecting only her face and causing difficulty of movement, particularly of her shoulders and knees. Physical examination revealed large, indurated, pearly white plaques in some areas (Fig. 1), and atrophic and wrinkled skin in other areas (Fig. 2). Additional tests, including complete blood count, biochemistry, antinuclear, anti-scl 70, and anticentromere antibodies, and serology for Borrelia burgdorferi, were normal or negative. Skin biopsy showed an atrophic epidermis with follicular occlusion and degeneration of the basal layer, edema of the papillary dermis, collagen with a hyalinized appearance, and a band-like lymphocytic infiltrate. The same sample also showed a thickened reticular dermis with thick and compact collagen bundles and a chronic inflammatory infiltrate (Fig. 3). Based on these clinical and histopathological findings, we made a diagnosis of morphea and generalized extragenital LSA and started treatment with oral psoralen–UV-A (PUVA). Given the poor response after 48 treatment sessions, oral prednisone, 20mg/d, was prescribed in combination with subcutaneous methotrexate, 15mg/wk, leading to a significant important in the induration and in mobility after 3 months of treatment, and it was possible to discontinue treatment after 10 months.
LSA and morphea are skin diseases of unknown etiology, though in many studies they are considered to be autoimmune diseases. They have been associated with infections, trauma, and, less frequently, with the administration of vaccines. We found 14 cases of vaccine-related morphea1–6 and a single case of LSA.7 However, only 2 of these cases were of generalized morphea (the vaccines implicated in these cases were bacillus Calmette-Guérin [BCG]5 and tetanus6), and neither occurred concomitantly with LSA.
The vaccine implicated in our patient was the inactivated surface antigen influenza vaccine, Chiroflu. This vaccine is recommended for seasonal influenza prophylaxis in persons over 65 years of age, health staff, pregnant women, individuals with respiratory or cardiac diseases, and immunodeficient patients.
Apart from the known adverse effects of vaccination, temporally or geographically related dermatoses have been reported after the intramuscular injection of vaccines, including granuloma annulare after hepatitis B, BCG, or tetanus vaccines, lichen planus after hepatitis B vaccine, and bullous pemphigoid after hepatitis B, DTP (diphtheria, tetanus, pertussis), or influenza vaccines. A temporal relationship was observed between influenza vaccination and the onset of morphea in our patient and, in addition, the first lesion developed at the site of injection of the vaccine. The role of this vaccine in the pathogenesis of morphea is poorly understood, but, as Torrelo et al.1 suggest, vaccines may stimulate an immune response targeting not only specific antigens of the vaccine but also other nonspecific antigens. An alternative proposal is that the trauma of the injection may cause endothelial damage and tissue hypoxia that could favor the development of morphea-type sclerosis. Trauma is associated with inflammation and the release of cytokines and growth factors, which contribute to the wound healing process at the site of vaccination and to the appearance of sclerosis or morphea.
The coexistence of morphea and LSA continues to be controversial, as some authors consider LSA to be a variant of morphea, with more superficial involvement, while others consider the diseases to be sufficiently different, both clinically and histopathologically, to be considered distinct diseases.8–10
A number of treatments have been attempted, including high-potency topical corticosteroids, systemic corticosteroids, methotrexate, and psoralen–UV-A PUVA, all with little efficacy.
We have described this case because of the limited number of reports of generalized morphea after vaccination. This is the first to occur after influenza vaccination.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: López SR, García YH, Díez SG, Allende BV. Morfea y liquen escleroatrófico extragenital generalizados tras vacuna antigripal. Actas Dermosifiliogr. 2018;109:86–88.