Skin involvement in Crohn disease (CD) occurs in up to 44% of the patients affected by this disease, depending on the series.1 From a pathophysiologic point of view, mucocutaneous lesions can be divided into specific lesions, caused by the same pathophysiologic mechanism as CD, reactive lesions, histologically different from the former and caused by cross-antigenicity between the skin and the digestive tract, and associated lesions, whose mechanism is not well understood.2 Metastatic CD presents with specific lesions. It was first described in 1965 and is the least common specific manifestation of CD. We present a case recently diagnosed in our unit.
The patient was a 35-year-old man with a 9-year history of CD, well controlled on infliximab at a dose of 5mg/kg every 8 weeks. He was referred from gastroenterology outpatients for an 8-week history of eczema on the anterior aspect of both thighs. The lesions had not responded to 5 weeks of topical therapy with clobetasol cream under an occlusive dressing.
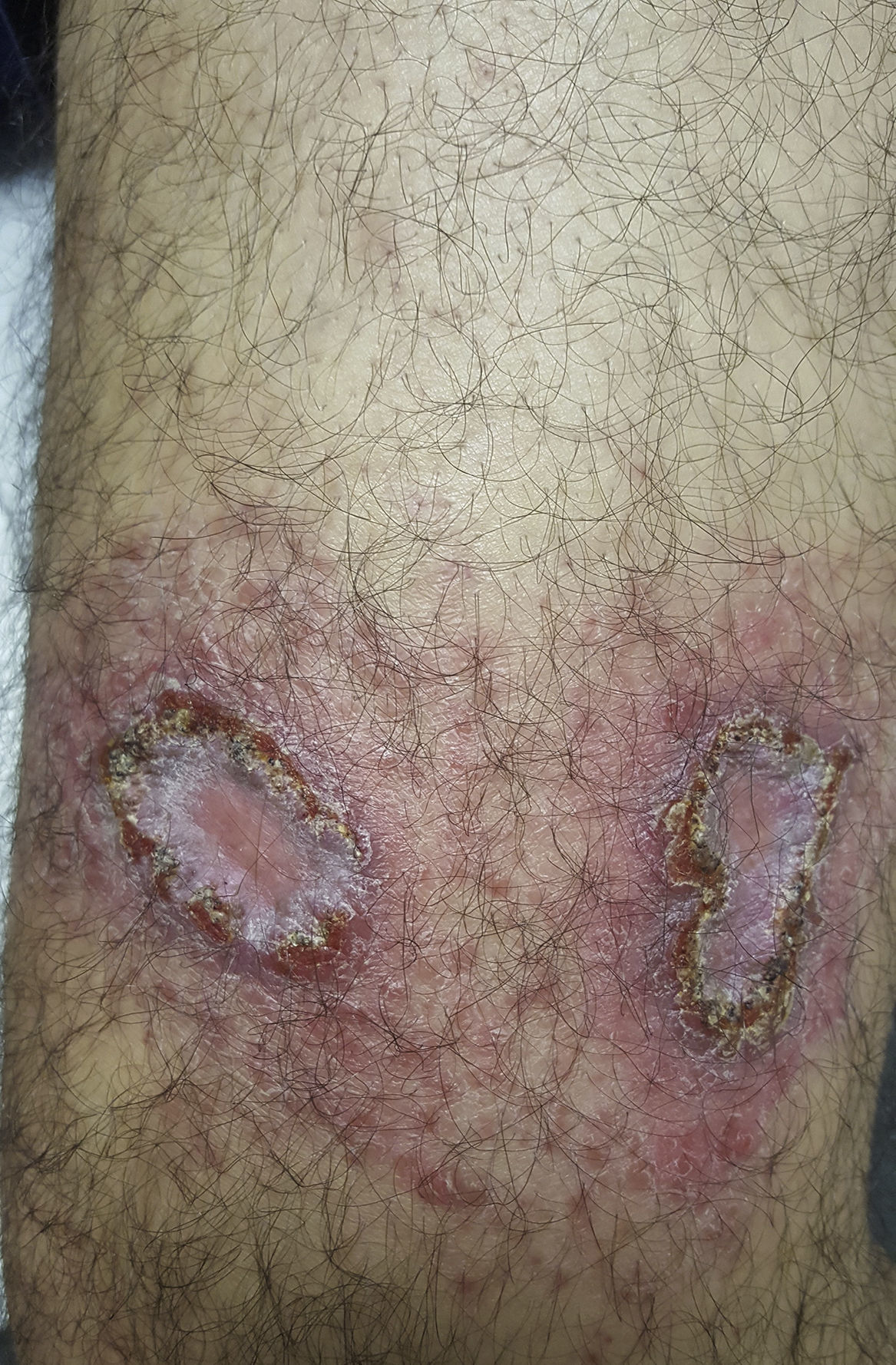
Physical examination revealed the presence of eczematous plaques of up to 5cm in diameter on the anterior aspect of both thighs. The lesions had a pale atrophic center with no ulceration and a slightly indurated erythematous border covered by a serous crust. There were no palpable locoregional lymph nodes (Figure 1). Additional tests (complete blood count, routine biochemistry, erythrocyte sedimentation rate, C-reactive protein, autoantibodies, circulating immune complexes, angiotensin converting enzyme, and 24-hour urinary calcium) were within normal limits and no abnormalities were observed on chest x-ray.
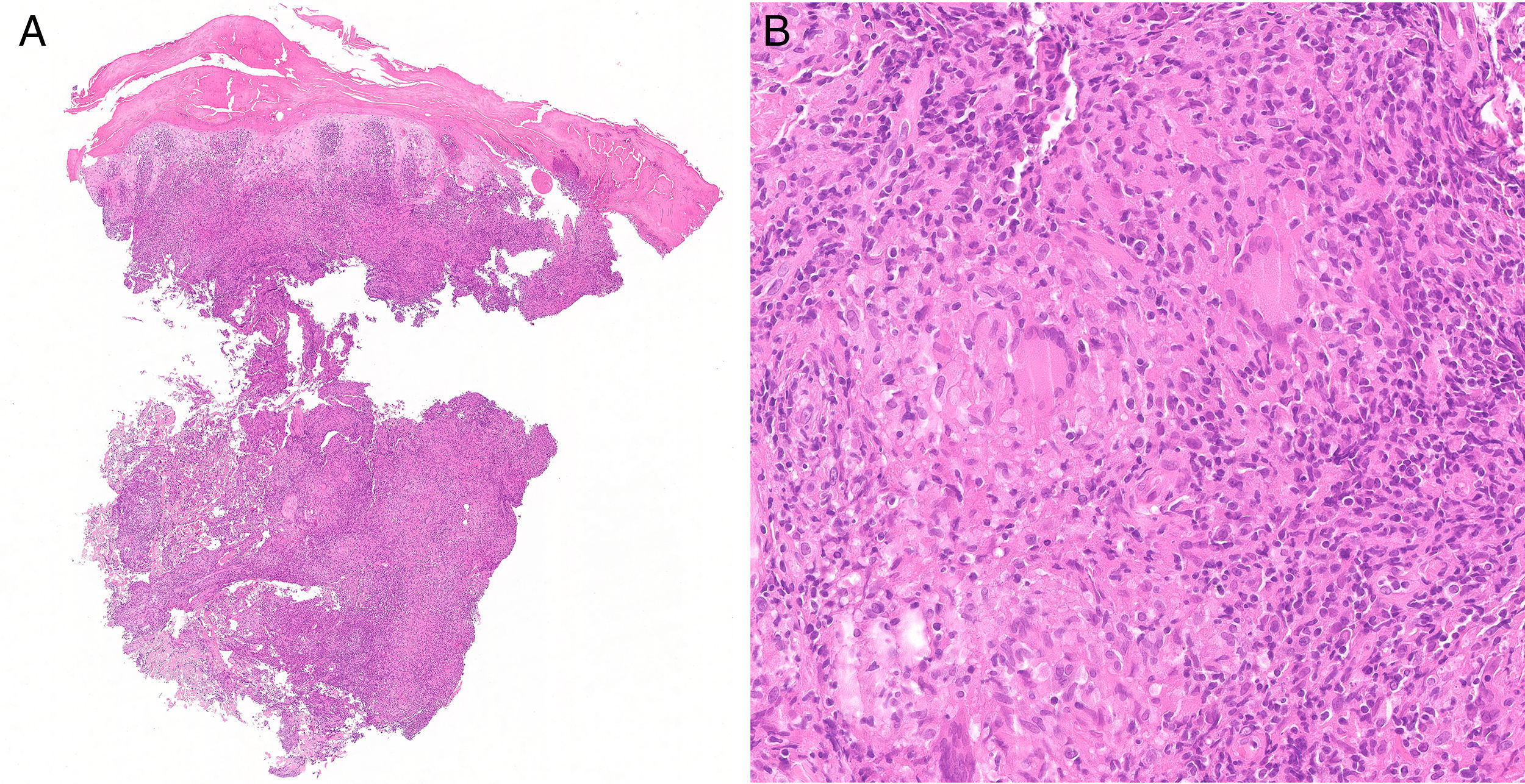
Histology revealed the presence of a chronic granulomatous inflammatory infiltrate in the dermis, with intense epithelial hyperplasia and foci of ulceration (Figure 2A). Noncaseating granulomas with multinucleated giant cells were visible at higher magnification, some with a perivascular distribution (Figure 2B). Stains (Ziehl-Neelsen and Grocott) performed to exclude mycobacterial and mycotic infections were negative.
Infliximab levels were normal (4μg/ml; normal target range, 3-10μg/ml) and antidrug antibody (ADA) levels were elevated (ADA, 15arbitrary units [AU]/ml; a value greater than 10AU/ml is considered positive).
Based on these findings, we made a diagnosis of metastatic CD. Treatment was started with prednisone at a dose of 0.5mg/kg/d, achieving moderate control of the cutaneous alterations. Changing the treatment of the patient's underlying disease to adalimumab at therapeutic doses for CD was suggested, and this led to the definitive resolution of the plaques for which the patient had consulted.
CD is currently considered to be 1 of the immune-mediated systemic diseases able to affect organs outside the digestive tract, such as the skin. Although there is no reliably demonstrated hypothesis, the presence of perivascular granulomas and circulating immune complexes points to phenomena of a vasculitic nature.
The global prevalence of metastatic CD is below 0.7%, with a slight female predominance, and it can affect the population with pediatric-onset CD.3 The clinical heterogeneity of the condition may be a cause of incorrect diagnosis or underdiagnosis.
Clinically, it presents as indurated erythematous plaques or nodules that may or may not be pigmented and that are sometimes ulcerated; there is no continuity between these lesions and the intestinal disease. The sites most frequently affected are the genital region,4 the skin folds (submammary, retroauricular, inguinal, and abdominal), and the lower limbs, though isolated or multiple lesions can arise anywhere on the skin. No relationship with activity of the intestinal disease is observed, though skin lesions appear to be more common among patients with involvement of the colon.
Histologically the lesions are characterized by the presence of noncaseating granulomas of epithelioid histiocytes and multinucleated cells in the papillary and reticular dermis, though no evidence has been found of concordance between the presence of granulomas on intestinal biopsy and their appearance in the skin. A perivascular lymphocytic inflammatory infiltrate is characteristic. Histology can be indistinguishable from cutaneous sarcoidosis.
In our case, due to the site of the lesions, the differential diagnosis included cellulitis, allergic contact dermatitis, pyoderma gangrenosum, and Wegener granulomatosis. From a pathologic viewpoint, the differential diagnosis included sarcoidosis, mycobacteriosis, and the deep mycoses.
No well-established action protocols have been drawn up and treatment is therefore prescribed in accordance with the patient's preference, the side effects, and the need for monitoring.2 The number of lesions and their site may allow us to use first-line topical therapies (corticosteroids or calcineurin inhibitors). If no response is achieved, systemic corticosteroids (prednisone, 0.5mg/kg), oral metronidazole (800-1500mg/d), conventional immunosuppressants (methotrexate, azathioprine, ciclosporin), or even biologics (infliximab, certolizumab, or adalimumab) may be required.5,6 Recently, case series have been published describing patients with pediatric-onset CD successfully treated with adalimumab after failure of initial therapy with infliximab, with follow-up for over a year, as occurred in the case we have presented.7
Please cite this article as: Ruiz-Villaverde R, Sánchez-Cano D, Perez-Lopez I, Aneiros-Fernández J. Enfermedad de Crohn metastásica. Actas Dermosifiliogr. 2017;108:171–172.