Infiltration of the skin by lymphoplasmacytic cells is very rare in Waldenström macroglobulinemia (WM).1
A 73-year-old man was diagnosed with WM in 2011 after investigation for anemia and monocytosis detected in a routine blood test. At the time of diagnosis, he presented massive bone marrow infiltration by lymphoplasmacytic cells, accounting for more than 45% of the total cell count, with λ/κ light chain restriction and a monoclonal immunoglobulin (Ig) M component in blood of 3.84g/dL (normal value<2.7g/dL). Study of the L265P mutation in exon 5 of gene MYD88 (3p22.2) was positive.
The metastatic work-up showed axillary, retroperitoneal, iliac, and inguinal lymphadenopathies and hepatosplenomegaly. The patient did not report symptoms suggestive of hyperviscosity, and adipose tissue biopsy was negative for amyloid. It was therefore decided to adopt a wait-and-see approach.
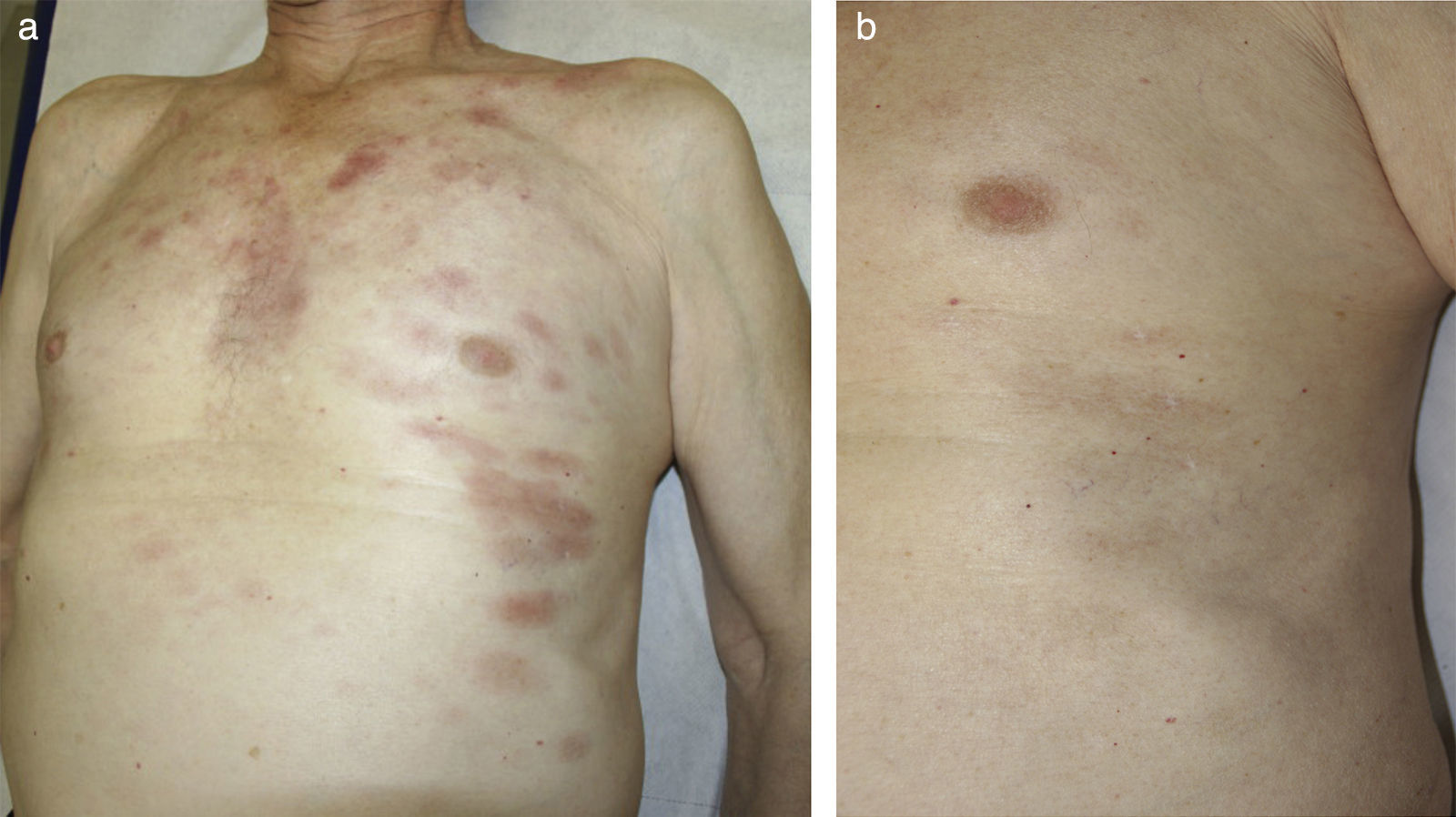
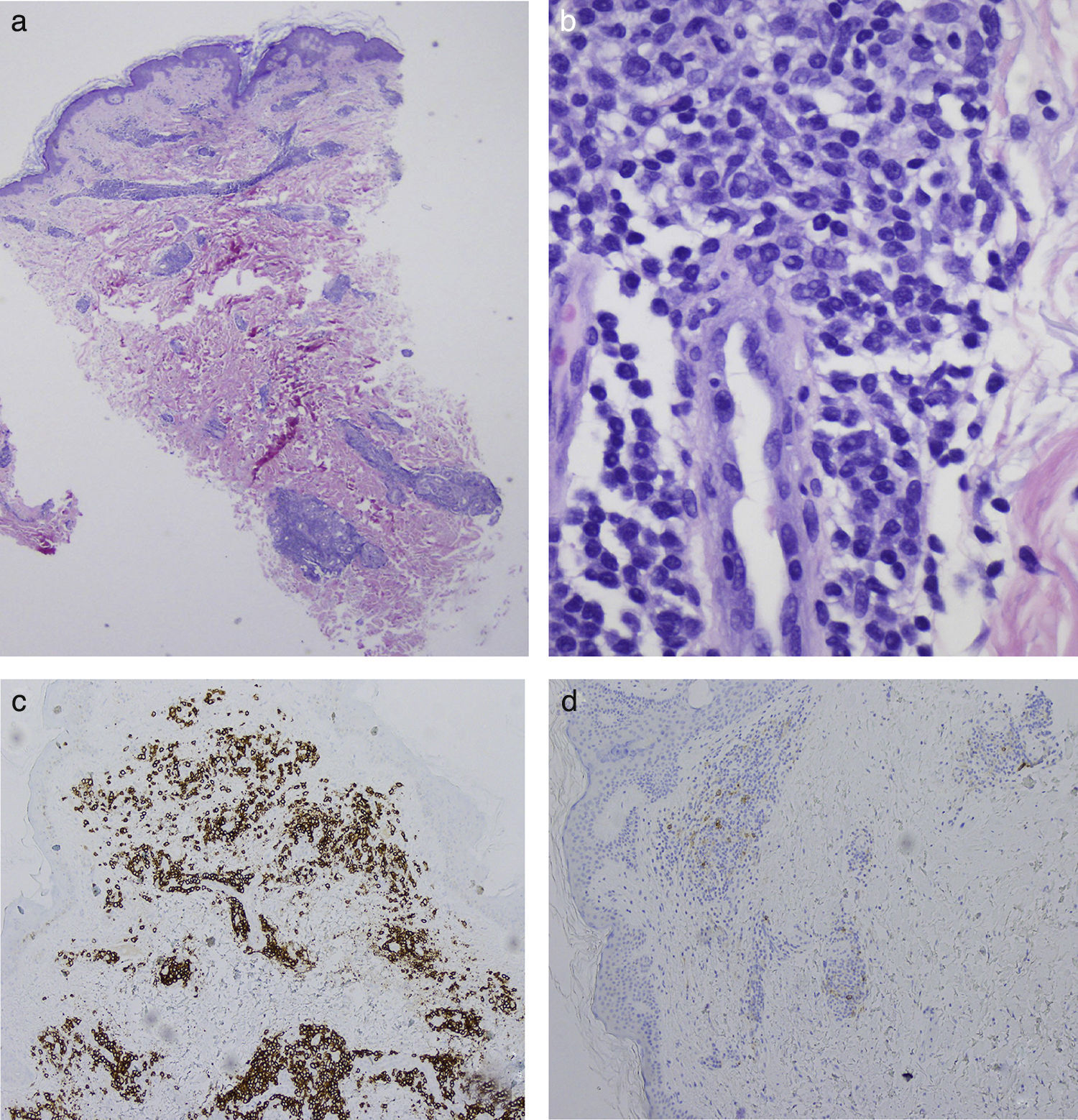
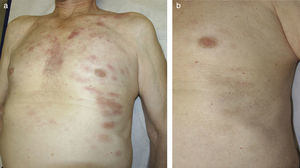
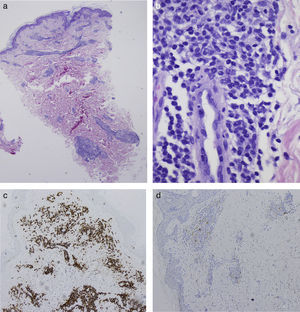
In September 2014, the patient was seen in dermatology for the appearance of asymptomatic erythematous plaques with mild desquamation on his upper trunk, back, and face (Fig. 1A). Histology revealed a lymphoplasmacytic infiltrate suggestive of B-cell lymphoma (Fig. 2, A and B), predominantly positive for CD20 (with a 2:1 ratio with CD3) and negative for CD38 and CD138 (Fig. 2, C and D). The few associated plasma cells did not show λ/κ light chain restriction. Genetic analysis identified positive monoclonal IgH (FR1 region) and biclonal IgH (FR3 region) rearrangements, confirming the diagnosis as cutaneous infiltration by lymphoplasmacytic lymphoma/WM. Immunohistochemistry for IgM was not performed on the skin biopsy because that study is not available at our hospital.
A, Perivascular lymphoplasmacytic infiltrates around the superficial and deep plexuses. Hematoxylin and eosin (H&E), original magnification×2. B, Periglandular lymphoplasmacytic infiltrates. H&E, original magnification×40. C, Positivity for CD20 in the lymphoplasmacytic infiltrate. CD20 immunohistochemical stain, original magnification×10. D, Negativity for CD38 in the neoplastic infiltrate. The few plasma cells that are present are not monoclonal cells and are positive for CD38. CD38 immunohistochemical stain, original magnification×10.
Clinical progression (asthenia, joint pain, weight loss, sweating) was observed during 2015, with progressive anemia (hemoglobin<10g/dL) and an increase in the size of the previously present lymph nodes. It was therefore decided to start systemic treatment with 6 cycles of rituximab plus bendamustine at standard doses. In addition, topical methylprednisolone was prescribed for the skin lesions. A month after completing the chemotherapy regimen, the skin lesions had disappeared. The patient reported an improvement in his general wellbeing, and blood test results (hemoglobin >12g/dL) and radiological findings had improved. At the time of writing, the patient has no general or cutaneous symptoms and continues on follow-up by the hematology department (Fig. 1B).
WM is a rare B-cell lymphoproliferative disorder of unknown etiology. It is characterized by a proliferation of lymphoplasmacytic cells in the bone marrow and a monoclonal IgM peak in peripheral blood.2
Skin involvement occurs in 5% of patients with WM. According to the classification by Libow et al., 2types of skin lesion can develop1,3: neoplastic lesions due to direct infiltration of the skin by the lymphoplasmacytic cells; and nonneoplastic lesions, secondary to the paraproteinemia. The nonneoplastic lesions are more common and are divided into 3 subtypes: those caused by a hyperviscosity syndrome (acral purpura, mucosal bleeding, peripheral edema); those associated with cryoglobulinemia (acral cyanosis, Raynaud phenomenon, cold hypersensitivity, livedo reticularis, leukocytoclastic vasculitis); and those related to the specific paraproteins (IgM bullous dermatosis, macroglobulinemia cutis, and erythematous papules associated with WM).
The neoplastic skin lesions, due to direct infiltration of the skin by the lymphoplasmacytic cells, are less common. Only around 20 cases have been reported in the literature, (Table 1)3–8 and their diagnosis in daily practice is very rare. They present clinically as mildly desquamating, slightly infiltrated erythematous plaques localized mainly symmetrically on the face and ears and also on the chest, flanks, and back.3–8 They can arise early in the course of the disease, but it should be stressed that their presence does not worsen the prognosis. Cells found in these skin lesions are from the lymphoplasmacytic cell line. They are positive for B-cell markers (CD19, CD20, CD22) and negative for plasma cell markers (CD38 and CD138).
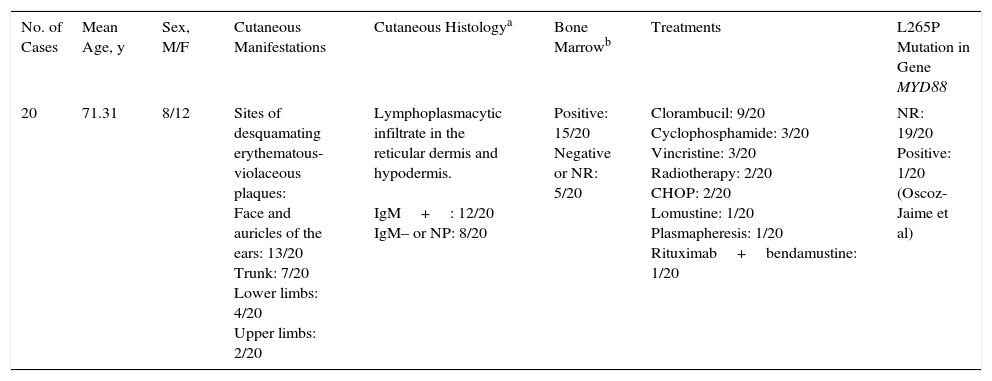
Summary of Cases of Skin Involvement by Waldenström Macroglobulinemia Reported in the Literature (See Additional Material).
| No. of Cases | Mean Age, y | Sex, M/F | Cutaneous Manifestations | Cutaneous Histologya | Bone Marrowb | Treatments | L265P Mutation in Gene MYD88 |
|---|---|---|---|---|---|---|---|
| 20 | 71.31 | 8/12 | Sites of desquamating erythematous-violaceous plaques: Face and auricles of the ears: 13/20 Trunk: 7/20 Lower limbs: 4/20 Upper limbs: 2/20 | Lymphoplasmacytic infiltrate in the reticular dermis and hypodermis. IgM+: 12/20 IgM– or NP: 8/20 | Positive: 15/20 Negative or NR: 5/20 | Clorambucil: 9/20 Cyclophosphamide: 3/20 Vincristine: 3/20 Radiotherapy: 2/20 CHOP: 2/20 Lomustine: 1/20 Plasmapheresis: 1/20 Rituximab+bendamustine: 1/20 | NR: 19/20 Positive: 1/20 (Oscoz-Jaime et al) |
Abbreviations: CHOP, cyclophosphamide+doxorubicin+vincristine+prednisone; F, female; M, male; NP, not performed; NR, not recorded or not performed.
The distinction between cases of lymphoplasmacytic lymphoma/WM and marginal zone lymphoma (MZL) with intense plasmacytic differentiation can be difficult in some cases because of the degree of clinical-pathologic overlap. Investigation of cyclin D1 (positive in MZL and negative in skin involvement by WM) and of MYD88 gene mutation (somatic mutation L265P is present in the majority of cases of WM9) and t11;18 translocation (present in MZL) can be used to make a definitive diagnosis.10
In multiple myeloma, the lesions analogous to the neoplastic lesions of WM are plasmacytomas. These present clinically as erythematous-violaceous or erythematous-yellowish plaques or nodules, and are the result of direct infiltration of the skin from underlying bony foci. Histology reveals a deep dermal and hypodermal infiltrate of plasma cells that are positive for CD38 and CD138 and negative for B-cell markers. In contrast to lesions due to infiltration by WM, the presence of plasmacytomas worsens the prognosis of multiple myeloma.
We draw attention to the peculiarities of our case because direct cutaneous infiltration by the lymphoproliferative disease is relatively rare, based on the literature, and does not lead to a worsening of the prognosis despite being a manifestation of disease progression.
Conflicts of InterestThe authors declare that they have no conflicts of interest.
Please cite this article as: Oscoz-Jaime S, Agulló-Pérez AD, Llanos-Chavarri C, Yanguas-Bayona JI. Infiltración cutánea por macroglobulinemia de Waldenström. Actas Dermosifiliogr. 2018;109:75–78.